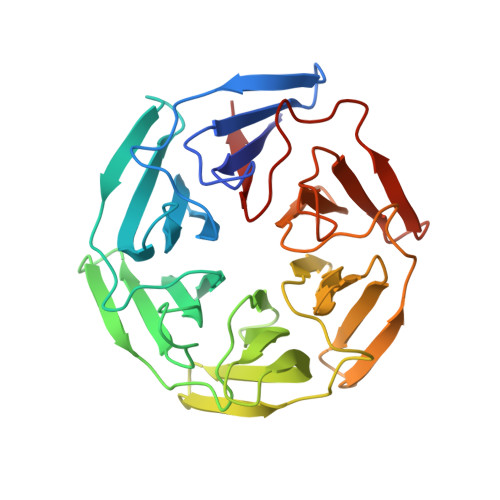
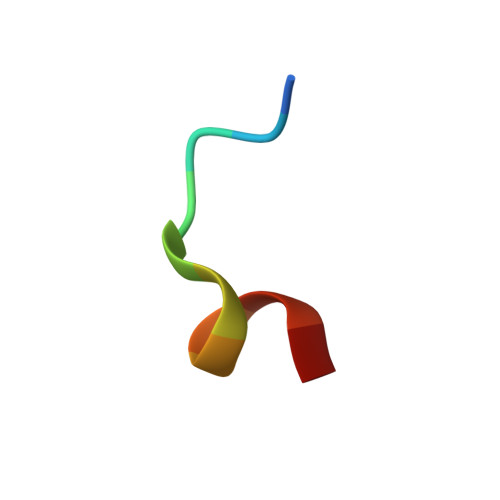Crystal structure of the human KLHL3 Kelch domain in complex with a WNK3 peptide
Chen, Z., Sorrell, F.J., Pinkas, D.M., Williams, E., Mathea, S., Goubin, S., von Delft, F., Arrowsmith, C.H., Edwards, A.M., Burgess-Brown, N., Bountra, C., Bullock, A.To be published.