Bartonella henselae BepA Fic domain in complex with its antitoxin homologue BiaA
Dietz, N., Schirmer, T.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Adenosine monophosphate-protein transferase | 301 | Bartonella henselae str. Houston-1 | Mutation(s): 0 Gene Names: bepA, BH13370 EC: 2.7.7 (PDB Primary Data), 2.7.7.108 (UniProt) | 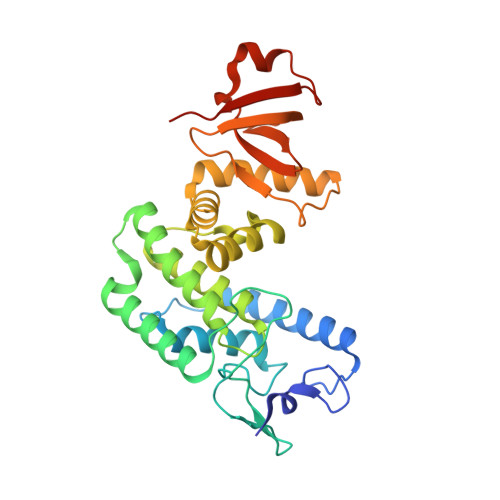 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q6G2A9 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Uncharacterized protein | 69 | Bartonella henselae str. Houston-1 | Mutation(s): 0 Gene Names: BH13360 | 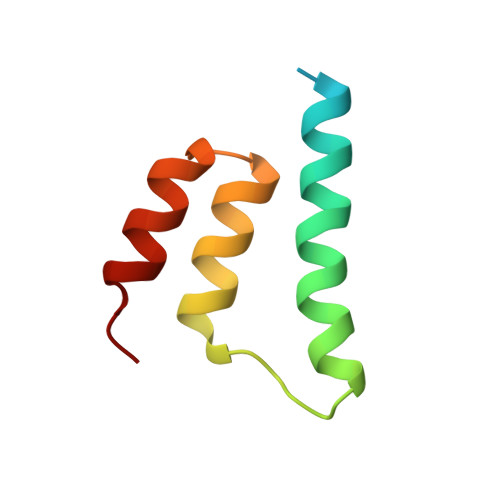 | |
UniProt | |||||
Find proteins for A0A0H3LZG7 (Bartonella henselae (strain ATCC 49882 / DSM 28221 / CCUG 30454 / Houston 1)) Explore A0A0H3LZG7 Go to UniProtKB: A0A0H3LZG7 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0A0H3LZG7 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| MG Download:Ideal Coordinates CCD File | C [auth A] | MAGNESIUM ION Mg JLVVSXFLKOJNIY-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 48.034 | α = 90 |
| b = 56.152 | β = 90 |
| c = 136.599 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| XDS | data reduction |
| Aimless | data scaling |
| PHASER | phasing |