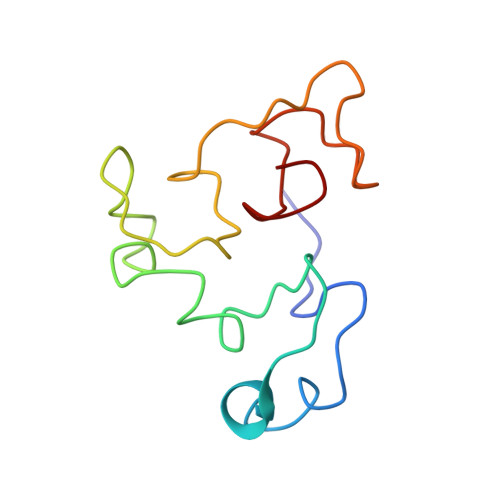
Structural Adaptation of a Protein to Increased Metal Stress: NMR Structure of a Marine Snail Metallothionein with an Additional Domain.
Baumann, C., Beil, A., Jurt, S., Niederwanger, M., Palacios, O., Capdevila, M., Atrian, S., Dallinger, R., Zerbe, O.(2017) Angew Chem Int Ed Engl 56: 4617-4622
- PubMed: 28332759 Search on PubMed
- DOI: https://doi.org/10.1002/anie.201611873
- Primary Citation Related Structures:
5ML1, 5MN3 - PubMed Abstract:
In this study, we present an NMR structure of the metallothionein (MT) from the snail Littorina littorea (LlMT) in complex with Cd 2+ . LlMT is capable of binding 9 Zn 2+ or 9 Cd 2+ ions. Sequence alignments with other snail MTs revealed that the protein is likely composed of three domains. The study revealed that the protein is divided into three individual domains, each of which folds into a single well-defined three-metal cluster. The central α2 and C-terminal β domains are positioned with a unique relative orientation. Two variants with longer and shorter linkers were investigated, which revealed that specific interdomain contacts only occurred with the wild-type linker. Moreover, a domain-swap mutant in which the highly similar α1 and α2 domains were exchanged was structurally almost identical. It is suggested that the expression of a three-domain MT confers an evolutionary advantage on Littorina littorea in terms of coping with Cd 2+ stress and adverse environmental conditions.
- Department of Chemistry, University of Zurich, Winterthurerstrasse 190, 8057, Zurich, Switzerland.
Organizational Affiliation: