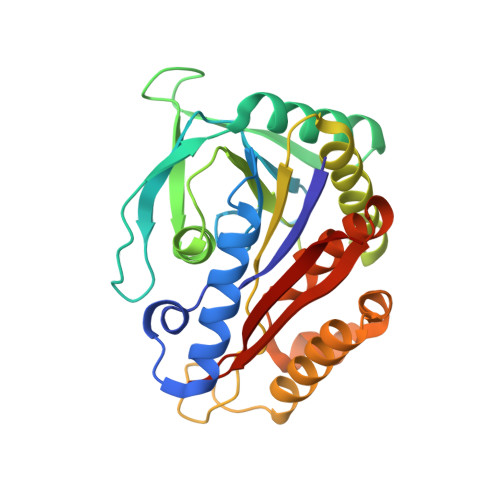
Crystal structure of the TK2203 protein from Thermococcus kodakarensis, a putative extradiol dioxygenase
Nishitani, Y., Simons, J.R., Kanai, T., Atomi, H., Miki, K.(2016) Acta Crystallogr F Struct Biol Commun 72: 427-433
- PubMed: 27303894 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1107/S2053230X16006920
- Primary Citation Related Structures:
5HEE - PubMed Abstract:
The TK2203 protein from the hyperthermophilic archaeon Thermococcus kodakarensis KOD1 (262 residues, 29 kDa) is a putative extradiol dioxygenase catalyzing the cleavage of C-C bonds in catechol derivatives. It contains three metal-binding residues, but has no significant sequence similarity to proteins for which structures have been determined. Here, the first crystal structure of the TK2203 protein was determined at 1.41 Å resolution to investigate its functional role. Structure analysis reveals that this protein shares the same fold and catalytic residues as other extradiol dioxygenases, strongly suggesting the same enzymatic activity. Furthermore, the important region contributing to substrate selectivity is discussed.
- Department of Chemistry, Graduate School of Science, Kyoto University, Sakyo-ku, Kyoto 606-8502, Japan.
Organizational Affiliation: