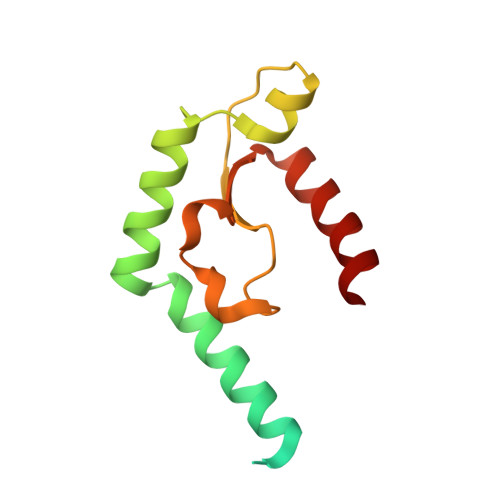
Structural and functional characterization of deep-sea thermophilic bacteriophage GVE2 HNH endonuclease
Zhang, L.K., Xu, D.D., Huang, Y.C., Zhu, X.Y., Rui, M.W., Wan, T., Zheng, X., Shen, Y.L., Chen, X.D., Ma, K.S., Gong, Y.(2017) Sci Rep 7: 42542-42542
- PubMed: 28211904 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/srep42542
- Primary Citation Related Structures:
5H0M, 5H0O - PubMed Abstract:
HNH endonucleases in bacteriophages play a variety of roles in the phage lifecycle as key components of phage DNA packaging machines. The deep-sea thermophilic bacteriophage Geobacillus virus E2 (GVE2) encodes an HNH endonuclease (GVE2 HNHE). Here, the crystal structure of GVE2 HNHE is reported. This is the first structural study of a thermostable HNH endonuclease from a thermophilic bacteriophage. Structural comparison reveals that GVE2 HNHE possesses a typical ββα-metal fold and Zn-finger motif similar to those of HNH endonucleases from other bacteriophages, apart from containing an extra α-helix, suggesting conservation of these enzymes among bacteriophages. Biochemical analysis suggests that the alanine substitutions of the conserved residues (H93, N109 and H118) in the HNH motif of GVE2 HNHE abolished 94%, 60% and 83% of nicking activity, respectively. Compared to the wild type enzyme, the H93A mutant displayed almost the same conformation while the N108A and H118A mutants had different conformations. In addition, the wild type enzyme was more thermostable than the mutants. In the presence of Mn 2+ or Zn 2+ , the wild type enzyme displayed distinct DNA nicking patterns. However, high Mn 2+ concentrations were needed for the N109A and H118A mutants to nick DNA while Zn 2+ inactivated their nicking activity.
- Marine Science &Technology Institute Department of Environmental Science and Engineering, Yangzhou University, China.
Organizational Affiliation: