Structural relationship of the tandem PDZ tandem from Grasp55 and its implication in the unconventional secretion pathway
Cartier-Michaud, A., Betzi, S., Shi, X., Shi, N., Morelli, X., Aurrand-Lions, M.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Golgi reassembly-stacking protein 2 | 235 | Mus musculus | Mutation(s): 0 Gene Names: Gorasp2, GOLPH6 | 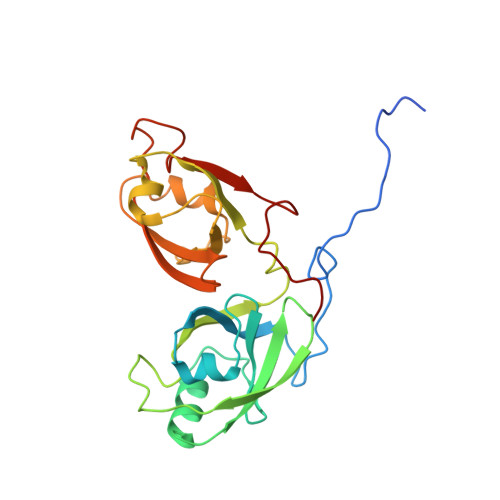 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q99JX3 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 132.312 | α = 90 |
| b = 132.312 | β = 90 |
| c = 221.055 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| PDB_EXTRACT | data extraction |
| PHASER | phasing |
| HKL-2000 | data reduction |
| xia2 | data scaling |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Natural Science Foundation of China | China | 31370738 |
| Campus France | France | PHC CAI YUANPEI 2013 - Project N26203WD |