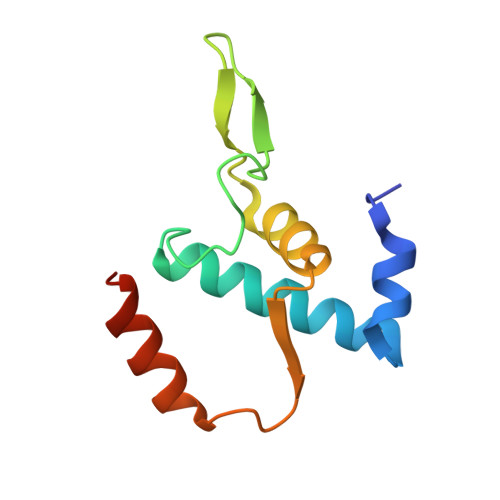
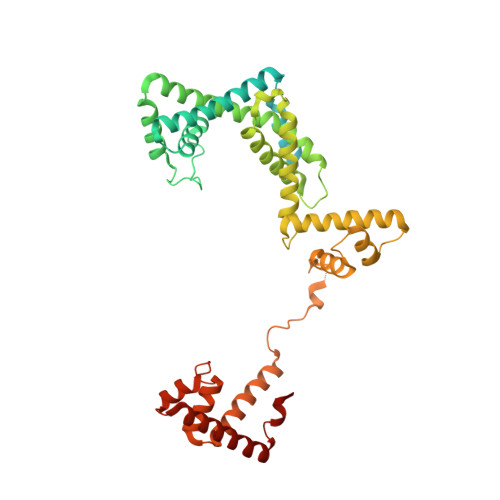
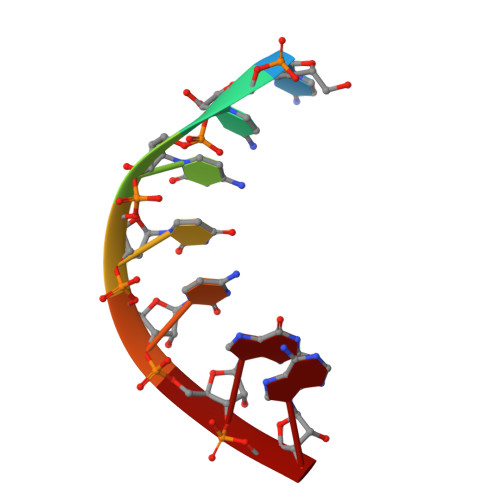
Multiplexed protein-DNA cross-linking: Scrunching in transcription start site selection.
Winkelman, J.T., Vvedenskaya, I.O., Zhang, Y., Zhang, Y., Bird, J.G., Taylor, D.M., Gourse, R.L., Ebright, R.H., Nickels, B.E.(2016) Science 351: 1090-1093
- PubMed: 26941320 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1126/science.aad6881
- Primary Citation Related Structures:
5E17, 5E18 - PubMed Abstract:
In bacterial transcription initiation, RNA polymerase (RNAP) selects a transcription start site (TSS) at variable distances downstream of core promoter elements. Using next-generation sequencing and unnatural amino acid-mediated protein-DNA cross-linking, we have determined, for a library of 4(10) promoter sequences, the TSS, the RNAP leading-edge position, and the RNAP trailing-edge position. We find that a promoter element upstream of the TSS, the "discriminator," participates in TSS selection, and that, as the TSS changes, the RNAP leading-edge position changes, but the RNAP trailing-edge position does not change. Changes in the RNAP leading-edge position, but not the RNAP trailing-edge position, are a defining hallmark of the "DNA scrunching" that occurs concurrent with RNA synthesis in initial transcription. We propose that TSS selection involves DNA scrunching prior to RNA synthesis.
- Department of Genetics, Rutgers University, Piscataway, NJ 08854, USA. Department of Chemistry and Chemical Biology, Rutgers University, Piscataway, NJ 08854, USA. Waksman Institute, Rutgers University, Piscataway, NJ 08854, USA. Department of Bacteriology, University of Wisconsin-Madison, Madison, WI 53705, USA.
Organizational Affiliation: