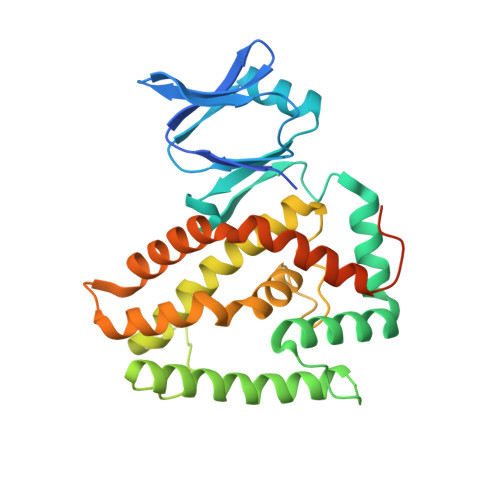
Structural and Mechanistic Basis of PAM-Dependent Spacer Acquisition in CRISPR-Cas Systems.
Wang, J., Li, J., Zhao, H., Sheng, G., Wang, M., Yin, M., Wang, Y.(2015) Cell 163: 840-853
- PubMed: 26478180 Search on PubMed
- DOI: https://doi.org/10.1016/j.cell.2015.10.008
- Primary Citation Related Structures:
5DLJ, 5DQT, 5DQU, 5DQZ - PubMed Abstract:
Bacteria acquire memory of viral invaders by incorporating invasive DNA sequence elements into the host CRISPR locus, generating a new spacer within the CRISPR array. We report on the structures of Cas1-Cas2-dual-forked DNA complexes in an effort toward understanding how the protospacer is sampled prior to insertion into the CRISPR locus. Our study reveals a protospacer DNA comprising a 23-bp duplex bracketed by tyrosine residues, together with anchored flanking 3' overhang segments. The PAM-complementary sequence in the 3' overhang is recognized by the Cas1a catalytic subunits in a base-specific manner, and subsequent cleavage at positions 5 nt from the duplex boundary generates a 33-nt DNA intermediate that is incorporated into the CRISPR array via a cut-and-paste mechanism. Upon protospacer binding, Cas1-Cas2 undergoes a significant conformational change, generating a flat surface conducive to proper protospacer recognition. Here, our study provides important structure-based mechanistic insights into PAM-dependent spacer acquisition.
- Key Laboratory of RNA Biology, Institute of Biophysics, Chinese Academy of Sciences, Beijing 100101, China; Beijing Key Laboratory of Noncoding RNA, Institute of Biophysics, Chinese Academy of Sciences, Beijing 100101, China.
Organizational Affiliation: