Crystal Structure of a Thermotoga maritima Hfq homolog
Patterson, J., Randolph, P.S., Mura, C.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| RNA-binding protein Hfq | 95 | Thermotoga maritima MSB8 | Mutation(s): 0 Gene Names: hfq, TM_0526 |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9WYZ6 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | ||||
| Molecule | Chains | Length | Organism | Image |
|---|---|---|---|---|
| RNA (5'-R(P*UP*UP*UP*UP*UP*U)-3') | M [auth N], N [auth O] | 6 | synthetic construct | 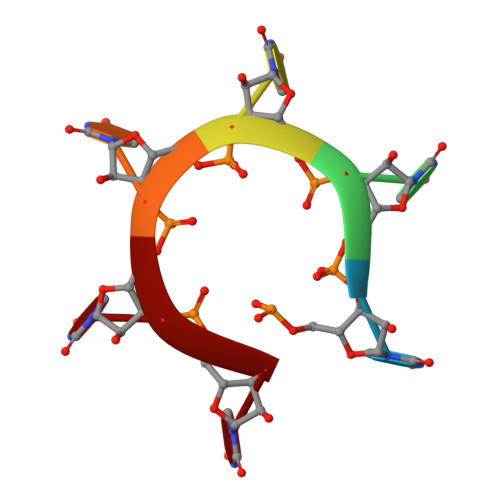 |
Sequence AnnotationsExpand | ||||
Reference Sequence | ||||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 39.08 | α = 90 |
| b = 133.5 | β = 90 |
| c = 206.18 | γ = 90 |
| Software Name | Purpose |
|---|---|
| XSCALE | data scaling |
| PHASER | phasing |
| PHENIX | refinement |
| PDB_EXTRACT | data extraction |
| XDS | data reduction |