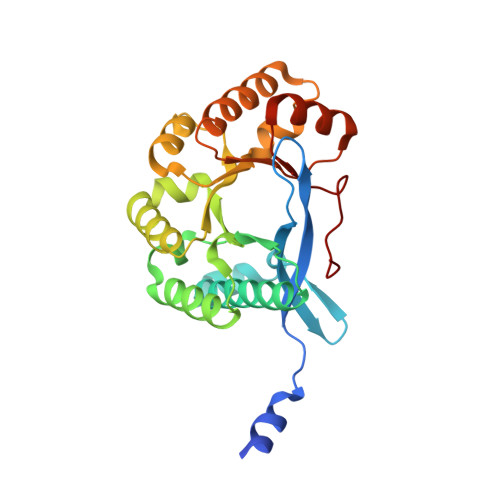
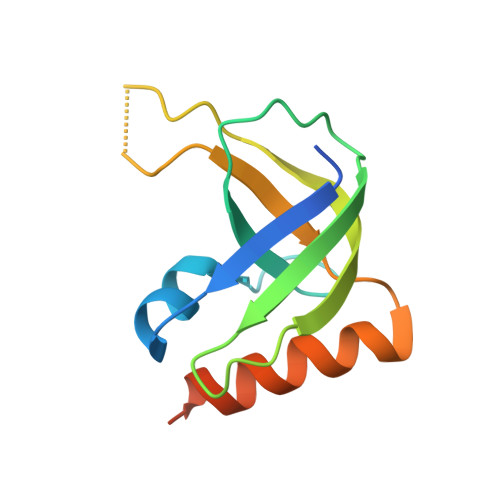
Structural polymorphism of c-di-GMP bound to an EAL domain and in complex with a type II PilZ-domain protein.
Chin, K.-H., Kuo, W.-T., Yu, Y.-J., Liao, Y.-T., Yang, M.T., Chou, S.-H.(2012) Acta Crystallogr D Biol Crystallogr 68: 1380-1392
- PubMed: 22993092 Search on PubMed
- DOI: https://doi.org/10.1107/S0907444912030594
- Primary Citation Related Structures:
4F3H, 4F48 - PubMed Abstract:
Cyclic di-GMP (c-di-GMP) is a novel secondary-messenger molecule that is involved in regulating a plethora of important bacterial activities through binding to an unprecedented array of effectors. Proteins with a canonical PilZ domain that bind c-di-GMP play crucial roles in regulating flagellum-based motility. In contrast, noncanonical type II PilZ domains that do not effectively bind c-di-GMP regulate twitching motility, which is dependent on type IV pili (T4P). Recent data indicate that T4P biogenesis is initiated via the interaction of a noncanonical type II PilZ protein with the GGDEF/EAL-domain protein FimX and the pilus motor protein PilB at high c-di-GMP concentrations. However, the molecular details of such interactions remain to be elucidated. In this manuscript, the first hetero-complex crystal structure between a type II PilZ protein and the EAL domain of the FimX protein (FimX(EAL)) from Xanthomonas campestris pv. campestris (Xcc) in the presence of c-di-GMP is reported. This work reveals two novel conformations of monomeric c-di-GMP in the XccFimX(EAL)-c-di-GMP and XccFimX(EAL)-c-di-GMP-XccPilZ complexes, as well as a unique interaction mode of a type II PilZ domain with FimX(EAL). These findings indicate that c-di-GMP is sufficiently flexible to adjust its conformation to match the corresponding recognition motifs of different cognate effectors. Together, these results represent a first step towards an understanding of how T4P biogenesis is controlled by c-di-GMP at the molecular level and also of the ability of c-di-GMP to bind to a wide variety of effectors.
- Agricultural Biotechnology Center, National Chung Hsing University, Taichung 40227, Taiwan.
Organizational Affiliation: