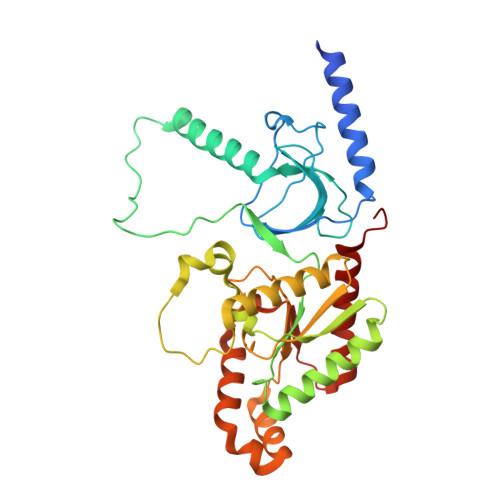
Structural and biophysical analyses of the skeletal dihydropyridine receptor beta subunit beta 1a reveal critical roles of domain interactions for stability.
Norris, N.C., Joseph, S., Aditya, S., Karunasekara, Y., Board, P.G., Dulhunty, A.F., Oakley, A.J., Casarotto, M.G.(2017) J Biol Chem 292: 8401-8411
- PubMed: 28351836
- DOI: https://doi.org/10.1074/jbc.M116.763896
- Primary Citation of Related Structures:
4ZW2 - PubMed Abstract:
Excitation-contraction (EC) coupling in skeletal muscle requires a physical interaction between the voltage-gated calcium channel dihydropyridine receptor (DHPR) and the ryanodine receptor Ca 2+ release channel. Although the exact molecular mechanism that initiates skeletal EC coupling is unresolved, it is clear that both the α 1 and β subunits of DHPR are essential for this process. Here, we employed a series of techniques, including size-exclusion chromatography-multi-angle light scattering, differential scanning fluorimetry, and isothermal calorimetry, to characterize various biophysical properties of the skeletal DHPR β subunit β 1a Removal of the intrinsically disordered N and C termini and the hook region of β 1a prevented oligomerization, allowing for its structural determination by X-ray crystallography. The structure had a topology similar to that of previously determined β isoforms, which consist of SH3 and guanylate kinase domains. However, transition melting temperatures derived from the differential scanning fluorimetry experiments indicated a significant difference in stability of ∼2-3 °C between the β 1a and β 2a constructs, and the addition of the DHPR α 1s I-II loop (α-interaction domain) peptide stabilized both β isoforms by ∼6-8 °C. Similar to other β isoforms, β 1a bound with nanomolar affinity to the α-interaction domain, but binding affinities were influenced by amino acid substitutions in the adjacent SH3 domain. These results suggest that intramolecular interactions between the SH3 and guanylate kinase domains play a role in the stability of β 1a while also providing a conduit for allosteric signaling events.
Organizational Affiliation:
John Curtin School of Medical Research, Australian National University, Canberra, Australian Capital Territory 2601, Australia.