`The Hunt for Serendipitous Allosteric Sites: Discovery of a novel allosteric inhibitor of the bacterial sialidase NanB
Rogers, G.W., Brear, P., Yang, L., Chen, A.S., Mitchell, J.B.O., Taylor, G.L., Westwood, N.J.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Sialidase B | 658 | Streptococcus pneumoniae TIGR4 | Mutation(s): 1 Gene Names: nanB, SP_1687 EC: 3.2.1.18 | 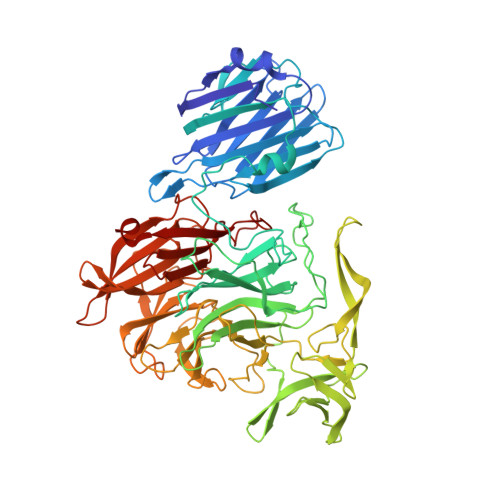 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q54727 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| 19D Download:Ideal Coordinates CCD File | G [auth A] | (1R,2R,3R,4R,5R,7R)-2-(acetylamino)-7-[(1R)-1,2-dihydroxyethyl]-4-fluoro-3-hydroxy-6,8-dioxabicyclo[3.2.1]octane-5-carboxylic acid C11 H16 F N O8 JCWSZUBTEOFKAD-DAXAGCIGSA-N |  | ||
| PO4 Download:Ideal Coordinates CCD File | F [auth A] | PHOSPHATE ION O4 P NBIIXXVUZAFLBC-UHFFFAOYSA-K |  | ||
| DMS Download:Ideal Coordinates CCD File | B [auth A], C [auth A], D [auth A], E [auth A] | DIMETHYL SULFOXIDE C2 H6 O S IAZDPXIOMUYVGZ-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 76.414 | α = 90 |
| b = 82.418 | β = 90 |
| c = 116.697 | γ = 90 |
| Software Name | Purpose |
|---|---|
| HKL-2000 | data reduction |
| PHASER | phasing |
| PHENIX | refinement |
| PDB_EXTRACT | data extraction |
| HKL-2000 | data scaling |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Biotechnology and Biological Sciences Research Council | United Kingdom | -- |
| SULSA | United Kingdom | -- |