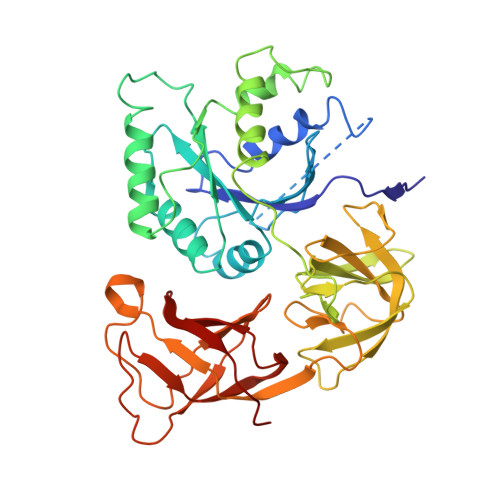
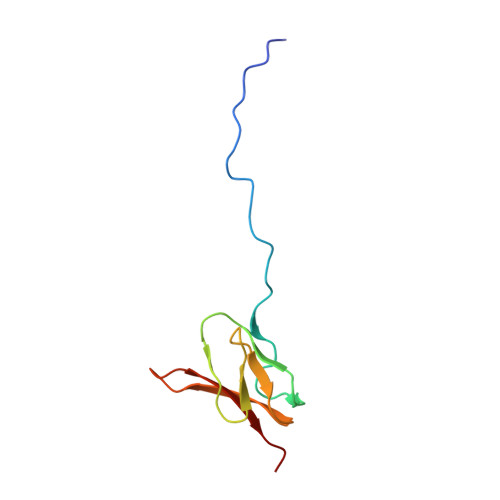
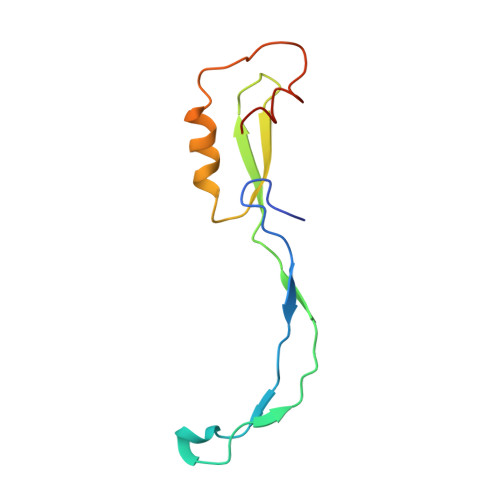
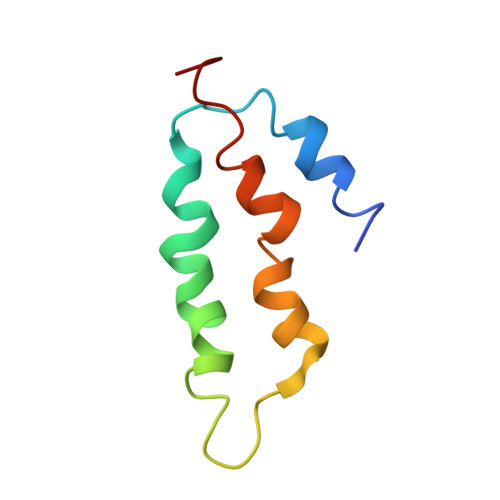
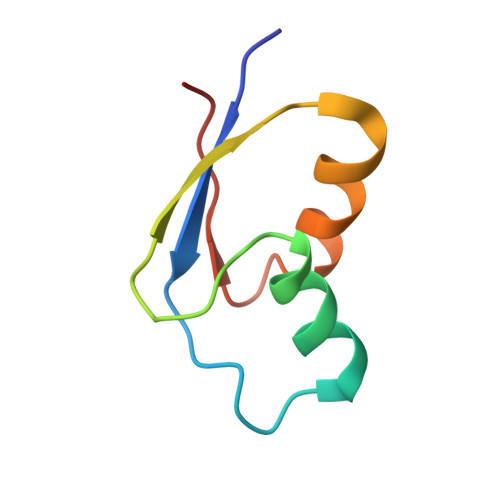
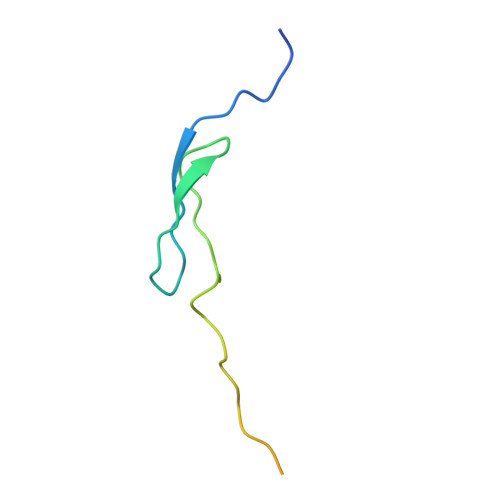
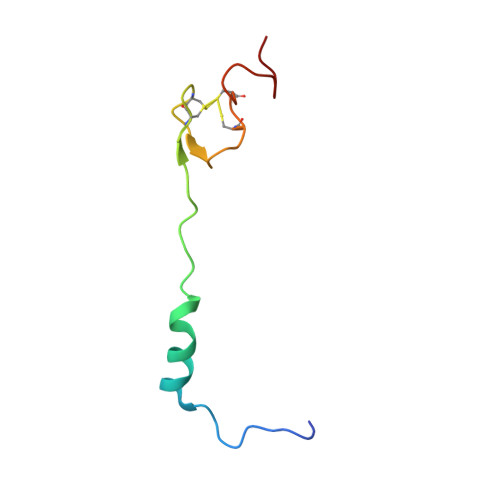
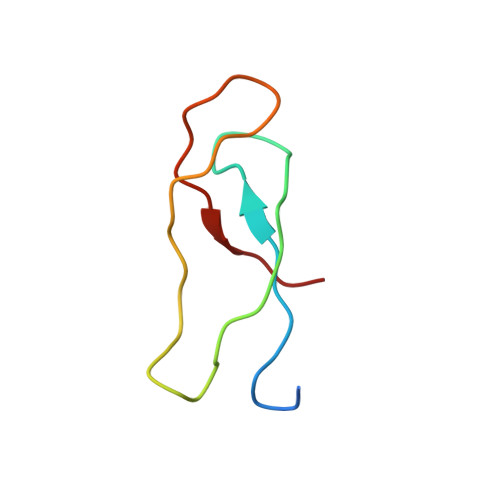
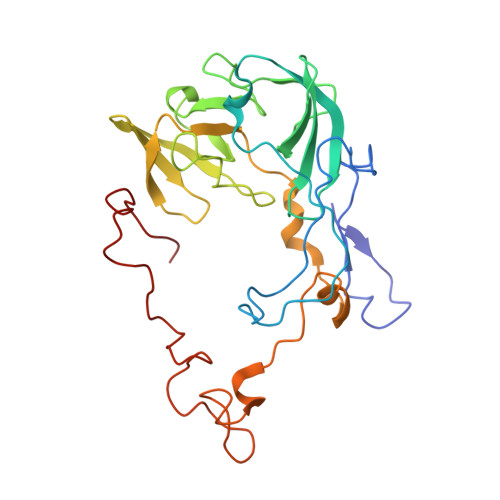
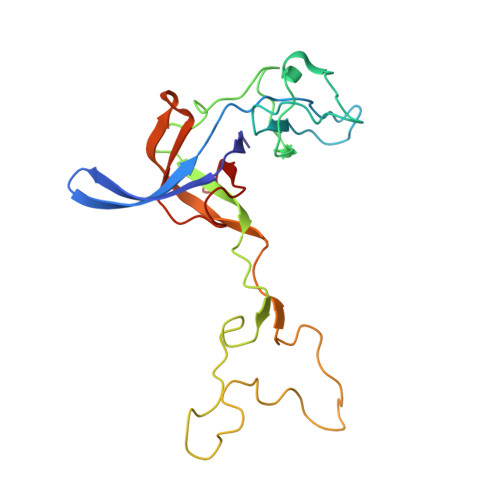
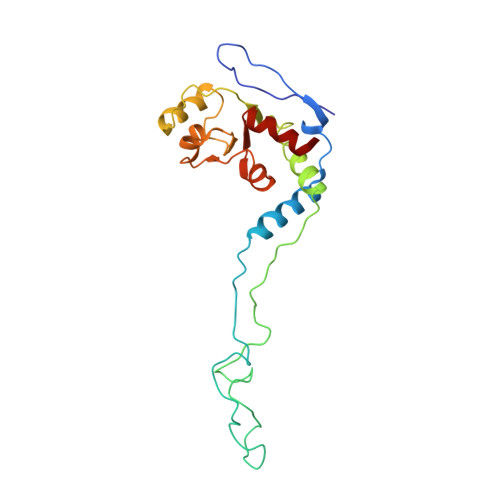
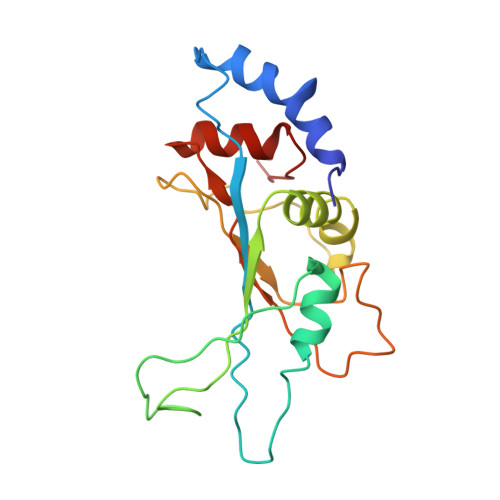
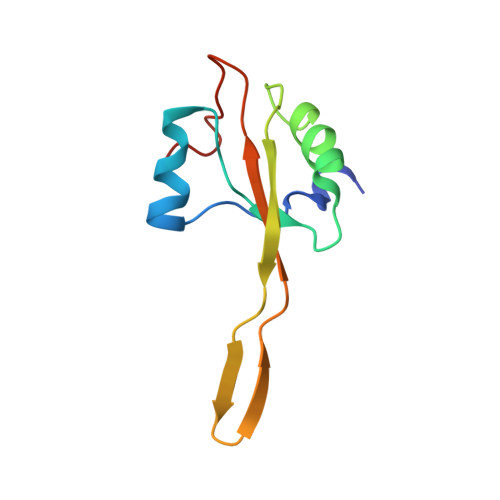
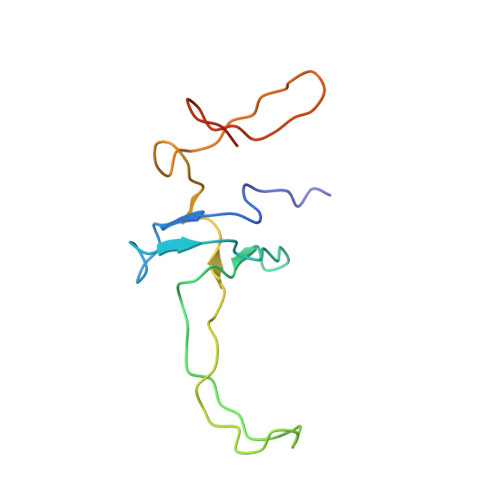
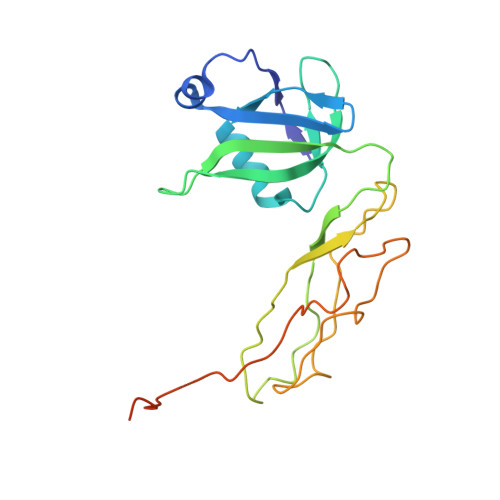
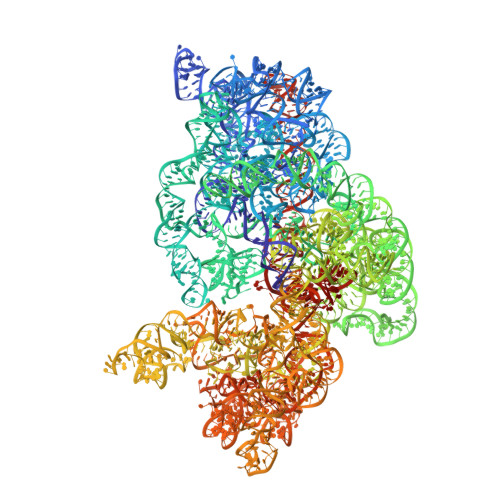
How Mutations in tRNA Distant from the Anticodon Affect the Fidelity of Decoding.
Schmeing, T.M., Voorhees, R.M., Kelley, A.C., Ramakrishnan, V.(2011) Nat Struct Mol Biol 18: 432
- PubMed: 21378964 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/nsmb.2003
- Primary Citation Related Structures:
4V5P, 4V5Q, 4V5R, 4V5S - PubMed Abstract:
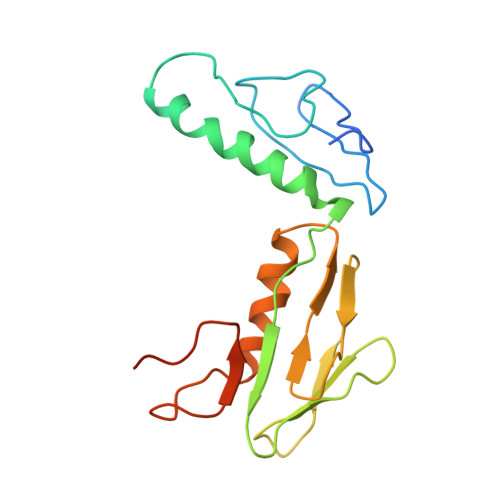
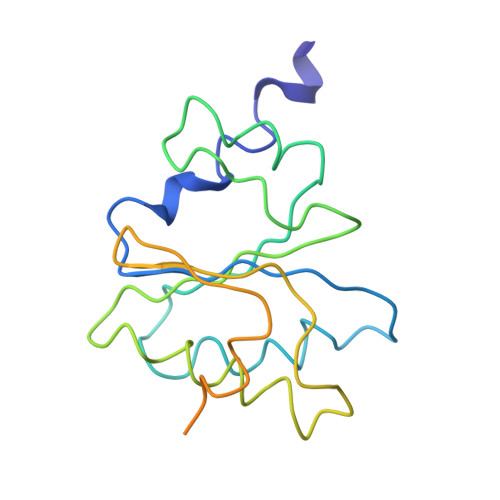
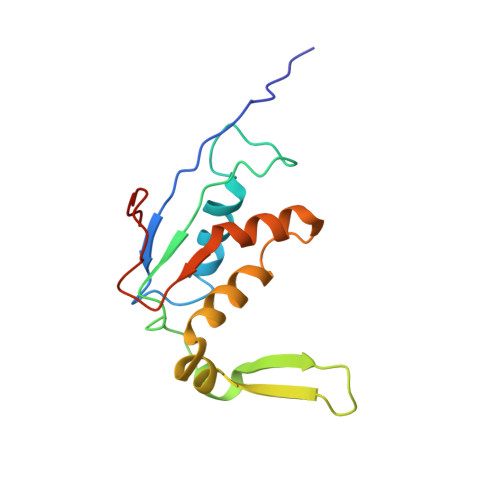
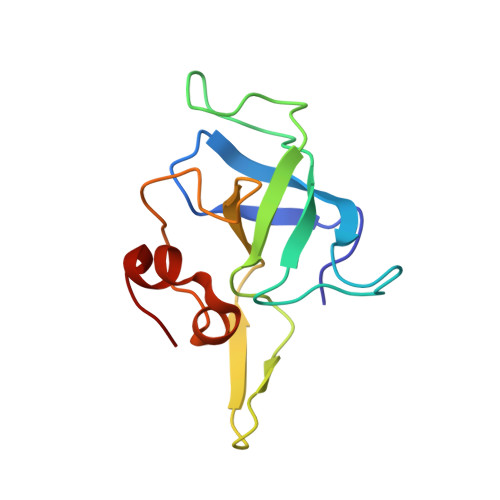
The ribosome converts genetic information into protein by selecting aminoacyl tRNAs whose anticodons base-pair to an mRNA codon. Mutations in the tRNA body can perturb this process and affect fidelity. The Hirsh suppressor is a well-studied tRNA(Trp) harboring a G24A mutation that allows readthrough of UGA stop codons. Here we present crystal structures of the 70S ribosome complexed with EF-Tu and aminoacyl tRNA (native tRNA(Trp), G24A tRNA(Trp) or the miscoding A9C tRNA(Trp)) bound to cognate UGG or near-cognate UGA codons, determined at 3.2-Å resolution. The A9C and G24A mutations lead to miscoding by facilitating the distortion of tRNA required for decoding. A9C accomplishes this by increasing tRNA flexibility, whereas G24A allows the formation of an additional hydrogen bond that stabilizes the distortion. Our results also suggest that each native tRNA will adopt a unique conformation when delivered to the ribosome that allows accurate decoding.
- MRC Laboratory of Molecular Biology, Cambridge, UK. martin.schmeing@mcgill.ca
Organizational Affiliation: