Structure-guided engineering of molinate hydrolase for the degradation of thiocarbamate pesticides.
Leite, J.P., Duarte, M., Paiva, A.M., Ferreira-da-Silva, F., Matias, P.M., Nunes, O.C., Gales, L.(2015) PLoS One 10: e0123430-e0123430
- PubMed: 25905461 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1371/journal.pone.0123430
- Primary Citation Related Structures:
4UB9 - PubMed Abstract:
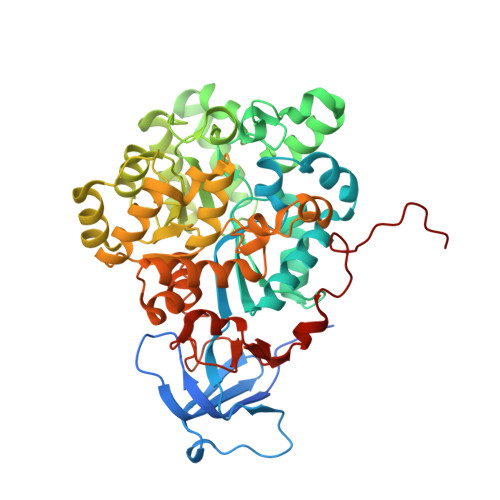
Molinate is a recalcitrant thiocarbamate used to control grass weeds in rice fields. The recently described molinate hydrolase, from Gulosibacter molinativorax ON4T, plays a key role in the only known molinate degradation pathway ending in the formation of innocuous compounds. Here we report the crystal structure of recombinant molinate hydrolase at 2.27 Å. The structure reveals a homotetramer with a single mononuclear metal-dependent active site per monomer. The active site architecture shows similarities with other amidohydrolases and enables us to propose a general acid-base catalysis mechanism for molinate hydrolysis. Molinate hydrolase is unable to degrade bulkier thiocarbamate pesticides such as thiobencarb which is used mostly in rice crops. Using a structural-based approach, we were able to generate a mutant (Arg187Ala) that efficiently degrades thiobencarb. The engineered enzyme is suitable for the development of a broader thiocarbamate bioremediation system.
- Instituto de Investigação e Inovação em Saúde, Universidade do Porto, Porto, Portugal; IBMC-Instituto de Biologia Molecular e Celular, Universidade do Porto, Porto, Portugal.
Organizational Affiliation: