Structure of the VIM-2 Metallo-beta-Lactamase in Complex with a Bisthiazolidine Inhibitor
Spencer, J.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Beta-lactamase class B VIM-2 | 242 | Pseudomonas aeruginosa | Mutation(s): 0 EC: 3.5.2.6 | 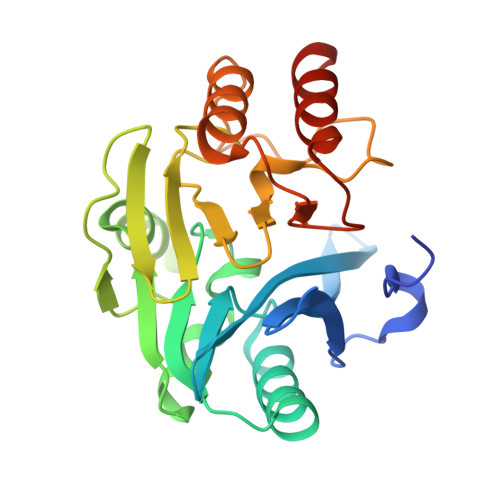 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9K2N0 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| 3C7 Download:Ideal Coordinates CCD File | F [auth A], G [auth A], H [auth B] | (3R,5R,7aS)-5-(sulfanylmethyl)tetrahydro[1,3]thiazolo[4,3-b][1,3]thiazole-3-carboxylic acid C7 H11 N O2 S3 ZTWVMVSSSBGFHH-JKUQZMGJSA-N |  | ||
| GOL Download:Ideal Coordinates CCD File | E [auth A], K [auth B], L [auth B], M [auth B] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| ZN Download:Ideal Coordinates CCD File | C [auth A], D [auth A], I [auth B], J [auth B] | ZINC ION Zn PTFCDOFLOPIGGS-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 52.888 | α = 90 |
| b = 61.404 | β = 100.87 |
| c = 68.918 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| XDS | data reduction |
| SCALA | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health | United States | R01AI100560 |