Effects of the regions adjacent to the RGD motif in disintegrins on their inhibitory activities and structures
Huang, C.H., Shiu, J.H., Chang, Y.T., Jeng, W.Y., Chuang, W.J.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Disintegrin rhodostomin | 68 | Calloselasma rhodostoma | Mutation(s): 5 Gene Names: RHOD | 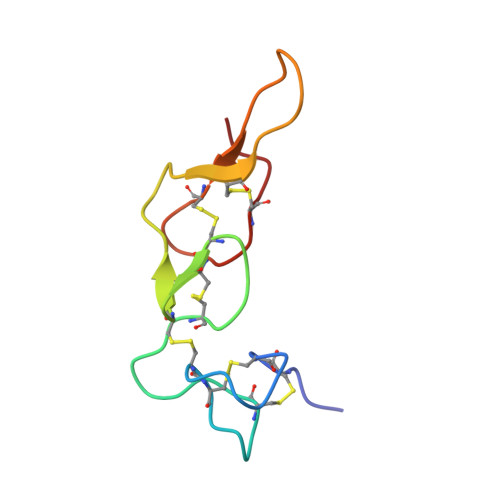 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P30403 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 18.083 | α = 90 |
| b = 79.986 | β = 91.3 |
| c = 39.497 | γ = 90 |
| Software Name | Purpose |
|---|---|
| HKL-2000 | data collection |
| PHASER | phasing |
| REFMAC | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |