Crystal Structure of the complex of a bimolecular human telomeric DNA with Coptisine
Ferraroni, M., Bazzicalupi, C., Gratteri, P., Bilia, A.R., Sissi, C.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | ||||
| Molecule | Chains | Length | Organism | Image |
|---|---|---|---|---|
| telomeric DNA | 12 | Homo sapiens | 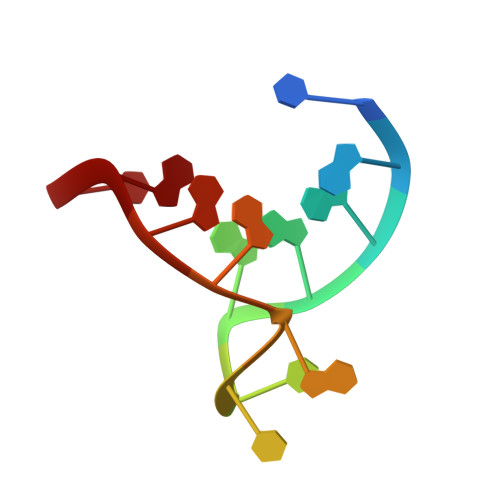 | |
Sequence AnnotationsExpand | ||||
Reference Sequence | ||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| KPT Download:Ideal Coordinates CCD File | E [auth A] | 6,7-dihydro[1,3]dioxolo[4,5-g][1,3]dioxolo[7,8]isoquino[3,2-a]isoquinolin-5-ium C19 H14 N O4 XYHOBCMEDLZUMP-UHFFFAOYSA-N |  | ||
| K Download:Ideal Coordinates CCD File | C [auth A], D [auth A] | POTASSIUM ION K NPYPAHLBTDXSSS-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 41.567 | α = 90 |
| b = 41.567 | β = 90 |
| c = 66.157 | γ = 90 |
| Software Name | Purpose |
|---|---|
| XDS | data reduction |
| XSCALE | data scaling |
| PDB_EXTRACT | data extraction |
| PHASER | phasing |
| REFMAC | refinement |