Atomic structure of the Immunophilin FKBP13-FK506 Complex: Insights into the Composite Binding Surface for Calcineurin
Schultz, L.W., Martin, P.K., Liang, J., Schreiber, S.L., Clardy, J.(1994) J Am Chem Soc 116: 3129-3130
Experimental Data Snapshot
(1994) J Am Chem Soc 116: 3129-3130
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Peptidyl-prolyl cis-trans isomerase FKBP2 | 142 | Homo sapiens | Mutation(s): 0 Gene Names: FKBP2, FKBP13 EC: 5.2.1.8 | 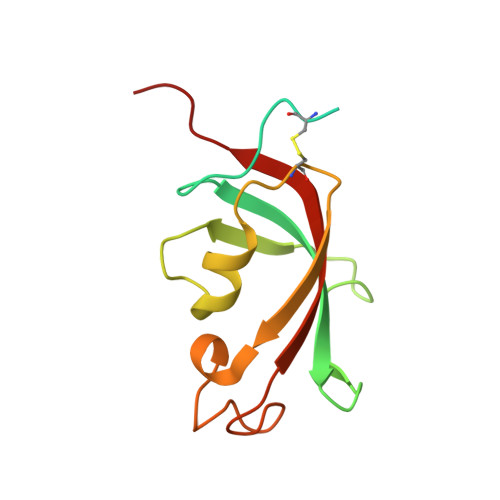 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P26885 GTEx: ENSG00000173486 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P26885 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| FK5 Download:Ideal Coordinates CCD File | C [auth A], D [auth B] | 8-DEETHYL-8-[BUT-3-ENYL]-ASCOMYCIN C44 H69 N O12 QJJXYPPXXYFBGM-LFZNUXCKSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 74.26 | α = 90 |
| b = 74.06 | β = 90 |
| c = 39.24 | γ = 90 |
| Software Name | Purpose |
|---|---|
| SDMS | data collection |
| X-PLOR | model building |
| X-PLOR | refinement |
| SDMS | data reduction |
| SCALEPACK | data scaling |
| X-PLOR | phasing |