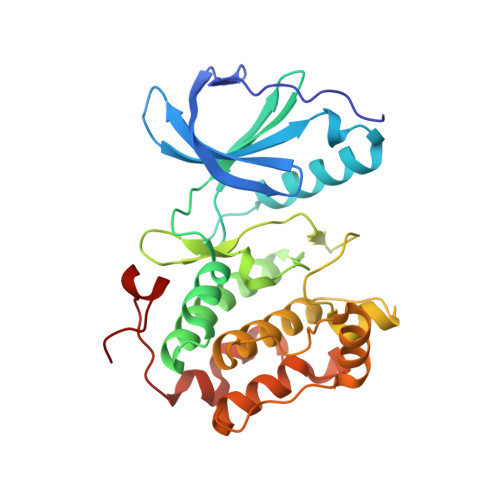
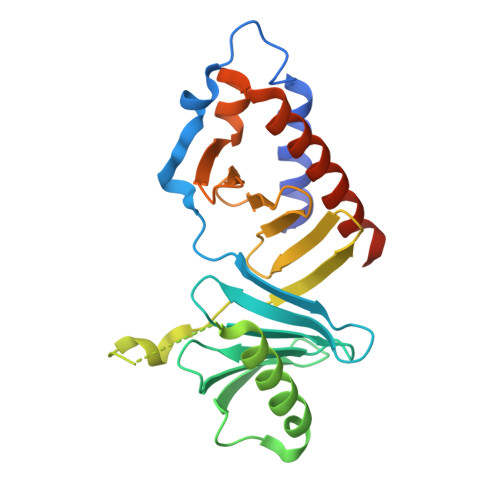
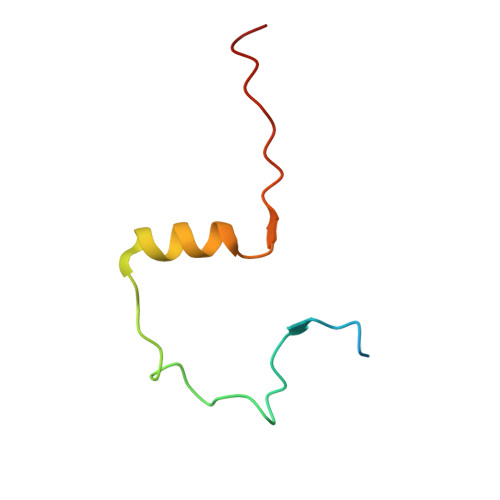
Structural basis for the inhibition of Polo-like kinase 1
Xu, J., Shen, C., Wang, T., Quan, J.(2013) Nat Struct Mol Biol 20: 1047-1053
- PubMed: 23893132 Search on PubMed
- DOI: https://doi.org/10.1038/nsmb.2623
- Primary Citation Related Structures:
4J7B - PubMed Abstract:
Polo-like kinase 1 (PLK1) is a master regulator of mitosis and is considered a potential drug target for cancer therapy. PLK1 is characterized by an N-terminal kinase domain (KD) and a C-terminal Polo-box domain (PBD). The KD and PBD are mutually inhibited, but the molecular mechanisms of the autoinhibition remain unclear. Here we report the 2.3-Å crystal structure of the complex of the Danio rerio KD and PBD together with a PBD-binding motif of Drosophila melanogaster microtubule-associated protein 205 (Map205(PBM)). The structure reveals that the PBD binds and rigidifies the hinge region of the KD in a distinct conformation from that of the phosphopeptide-bound PBD. This structure provides a framework for understanding the autoinhibitory mechanisms of PLK1 and also sheds light on the activation mechanisms of PLK1 by phosphorylation or phosphopeptide binding.
- Laboratory of Chemical Genomics, School of Chemical Biology and Biotechnology, Peking University Shenzhen Graduate School, Shenzhen, China.
Organizational Affiliation: