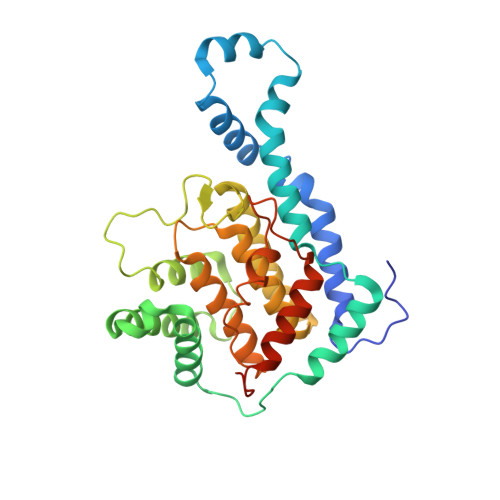
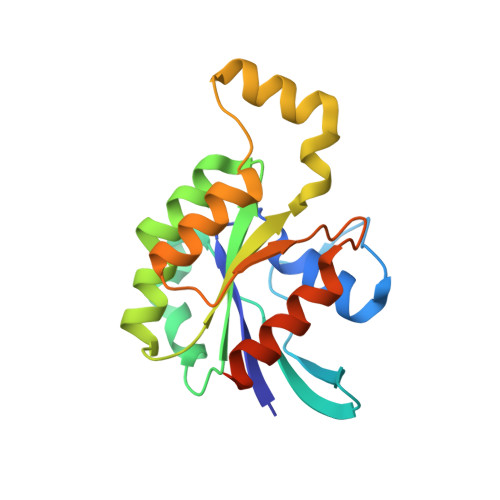
Structural basis of Fic-mediated adenylylation.
Xiao, J., Worby, C.A., Mattoo, S., Sankaran, B., Dixon, J.E.(2010) Nat Struct Mol Biol 17: 1004-1010
- PubMed: 20622875 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/nsmb.1867
- Primary Citation Related Structures:
3N3U, 4ITR - PubMed Abstract:
The Fic family of adenylyltransferases, defined by a core HPFx(D/E)GN(G/K)R motif, consists of over 2,700 proteins found in organisms from bacteria to humans. The immunoglobulin-binding protein A (IbpA) from the bacterial pathogen Histophilus somni contains two Fic domains that adenylylate the switch1 tyrosine residue of Rho-family GTPases, allowing the bacteria to subvert host defenses. Here we present the structure of the second Fic domain of IbpA (IbpAFic2) in complex with its substrate, Cdc42. IbpAFic2-bound Cdc42 mimics the GDI-bound state of Rho GTPases, with both its switch1 and switch2 regions gripped by IbpAFic2. Mutations disrupting the IbpAFic2-Cdc42 interface impair adenylylation and cytotoxicity. Notably, the switch1 tyrosine of Cdc42 is adenylylated in the structure, providing the first structural view for this post-translational modification. We also show that the nucleotide-binding mechanism is conserved among Fic proteins and propose a catalytic mechanism for this recently discovered family of enzymes.
- Department of Pharmacology, Howard Hughes Medical Institute, University of California, San Diego, La Jolla, California, USA.
Organizational Affiliation: