Regulation of a Viral Proteinase by a Peptide and DNA in One-dimensional Space: III. ATOMIC RESOLUTION STRUCTURE OF THE NASCENT FORM OF THE ADENOVIRUS PROTEINASE.
Baniecki, M.L., McGrath, W.J., Mangel, W.F.(2013) J Biological Chem 288: 2081-2091
- PubMed: 23043139 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M112.407429
- Primary Citation Related Structures:
4EKF - PubMed Abstract:
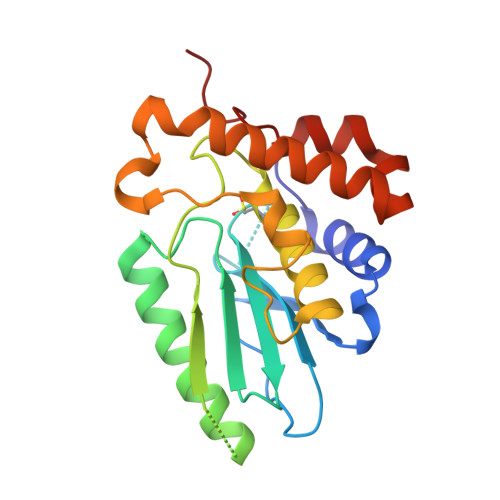
The adenovirus proteinase (AVP), the first member of a new class of cysteine proteinases, is essential for the production of infectious virus, and here we report its structure at 0.98 Å resolution. AVP, initially synthesized as an inactive enzyme, requires two cofactors for maximal activity: pVIc, an 11-amino acid peptide, and the viral DNA. Comparison of the structure of AVP with that of an active form, the AVP-pVIc complex, reveals why AVP is inactive. Both forms have an α + β fold; the major structural differences between them lie in the β-sheet domain. In AVP-pVIc, the general base His-54 Nδ1 is 3.9 Å away from the Cys-122 Sγ, thereby rendering it nucleophilic. In AVP, however, His-54 Nδ1 is 7.0 Å away from Cys-122 Sγ, too far away to be able to abstract the proton from Cys-122. In AVP-pVIc, Tyr-84 forms a cation-π interaction with His-54 that should raise the pK(a) of His-54 and freeze the imidazole ring in the place optimal for forming an ion pair with Cys-122. In AVP, however, Tyr-84 is more than 11 Å away from its position in AVP-pVIc. Based on the structural differences between AVP and AVP-pVIc, we present a model that postulates that activation of AVP by pVIc occurs via a 62-amino acid-long activation pathway in which the binding of pVIc initiates contiguous conformational changes, analogous to falling dominos. There is a common pathway that branches into a pathway that leads to the repositioning of His-54 and another pathway that leads to the repositioning of Tyr-84.
- Biology Department, Brookhaven National Laboratory, Upton, New York 11973, USA.
Organizational Affiliation: