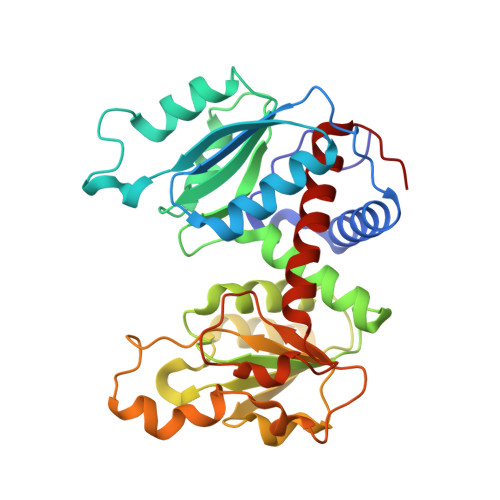
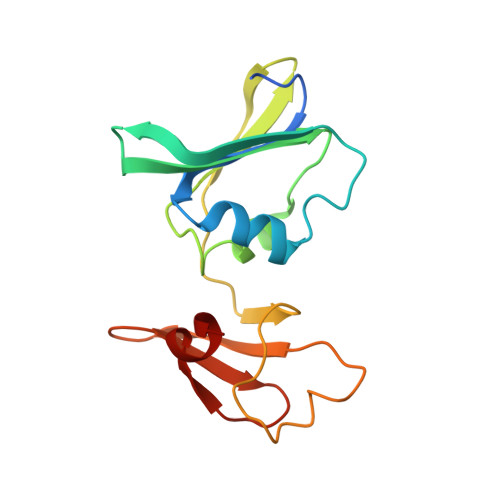
Trapping and structure determination of an intermediate in the allosteric transition of aspartate transcarbamoylase.
Guo, W., West, J.M., Dutton, A.S., Tsuruta, H., Kantrowitz, E.R.(2012) Proc Natl Acad Sci U S A 109: 7741-7746
- PubMed: 22547808 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.1119683109
- Primary Citation Related Structures:
4E2F - PubMed Abstract:
X-ray crystallography and small-angle X-ray scattering (SAXS) in solution have been used to show that a mutant aspartate transcarbamoylase exists in an intermediate quaternary structure between the canonical T and R structures. Additionally, the SAXS data indicate a pH-dependent structural alteration consistent with either a pH-induced conformational change or a pH-induced alteration in the T to R equilibrium. These data indicate that this mutant is not a model for the R state, as has been proposed, but rather represents the enzyme trapped along the path of the allosteric transition between the T and R states.
- Department of Chemistry, Merkert Chemistry Center, Boston College, Chestnut Hill, MA 02467, USA.
Organizational Affiliation: