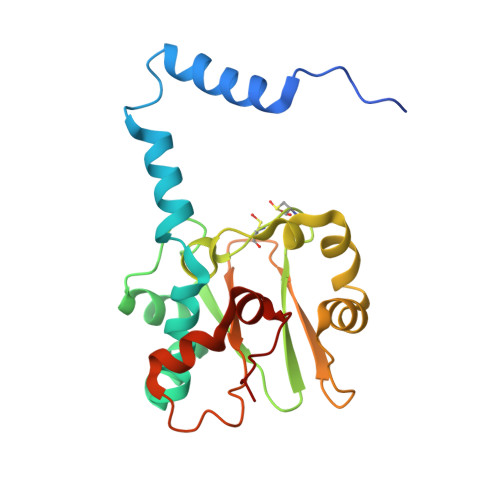
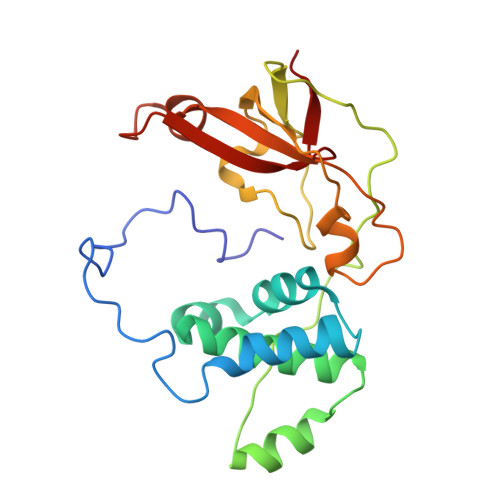
Time-Resolved Crystallography of the Reaction Intermediate of Nitrile Hydratase: Revealing a Role for the Cysteinesulfenic Acid Ligand as a Catalytic Nucleophile.
Yamanaka, Y., Kato, Y., Hashimoto, K., Iida, K., Nagasawa, K., Nakayama, H., Dohmae, N., Noguchi, K., Noguchi, T., Yohda, M., Odaka, M.(2015) Angew Chem Int Ed Engl 54: 10763-10767
- PubMed: 26333053 Search on PubMed
- DOI: https://doi.org/10.1002/anie.201502731
- Primary Citation Related Structures:
3WVD, 3WVE, 3X20, 3X24, 3X25, 3X26 - PubMed Abstract:
The reaction mechanism of nitrile hydratase (NHase) was investigated using time-resolved crystallography of the mutant NHase, in which βArg56, strictly conserved and hydrogen bonded to the two post-translationally oxidized cysteine ligands, was replaced by lysine, and pivalonitrile was the substrate. The crystal structures of the reaction intermediates were determined at high resolution (1.2-1.3 Å). In combination with FTIR analyses of NHase following hydration in H2 (18) O, we propose that the metal-coordinated substrate is nucleophilically attacked by the O(SO(-) ) atom of αCys114-SO(-) , followed by nucleophilic attack of the S(SO(-) ) atom by a βArg56-activated water molecule to release the product amide and regenerate αCys114-SO(-) .
- Department of Biotechnology and Life Science, Graduate School of Technology, Tokyo University of Agriculture and Technology, Koganei, Tokyo 184-8588 (Japan).
Organizational Affiliation: