Crystal structure of anti-Prostaglandin E2 Fab fragment with Prostaglandin E2
Sugahara, M., Ago, H., Saino, H., Miyano, M., Kurahashi, Y., Aoyama, S., Takehira, M., Yutani, K., Yamamoto, S.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| mAb Fab H fragment | 218 | Mus musculus | Mutation(s): 0 | 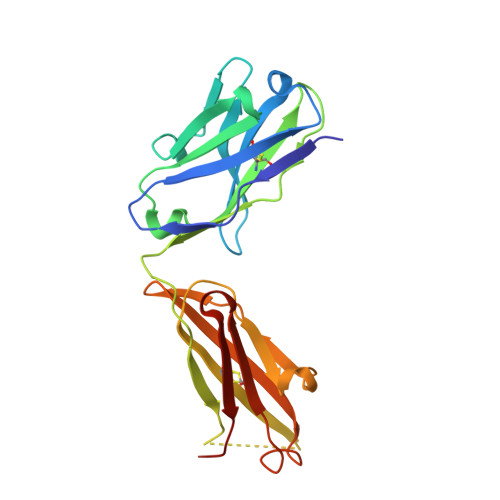 | |
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| mAb Fab L fragment | 217 | Mus musculus | Mutation(s): 0 | 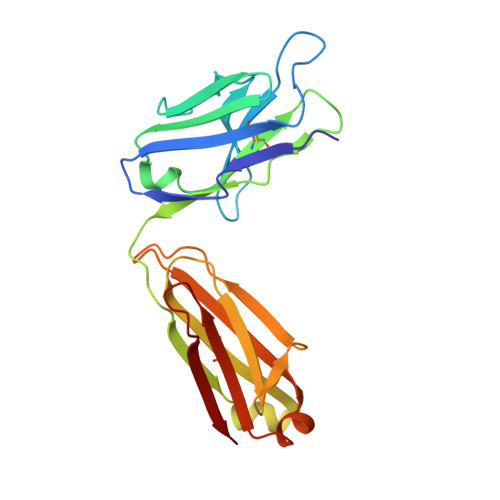 | |
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| XPG Download:Ideal Coordinates CCD File | C [auth B] | 7-[(1R,3R)-3-hydroxy-2-[(1E,3S)-3-hydroxyoct-1-en-1-yl]-5-oxocyclopentyl]heptanoic acid C20 H34 O5 GMVPRGQOIOIIMI-DWKJAMRDSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 70.46 | α = 90 |
| b = 82.03 | β = 90 |
| c = 82.31 | γ = 90 |
| Software Name | Purpose |
|---|---|
| BSS | data collection |
| MOLREP | phasing |
| PHENIX | refinement |
| MOSFLM | data reduction |
| SCALA | data scaling |