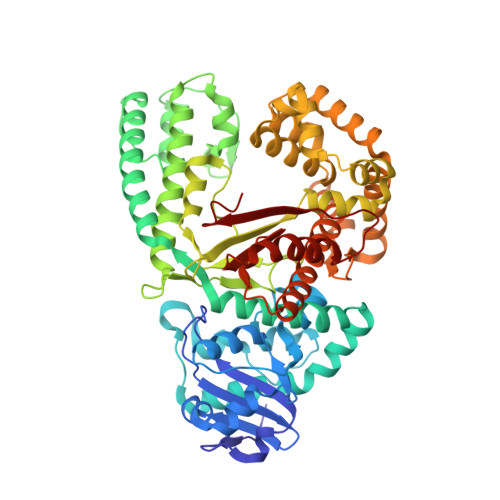
The Mechanism of the Translocation Step in DNA Replication by DNA Polymerase I: A Computer Simulation Analysis.
Golosov, A.A., Warren, J.J., Beese, L.S., Karplus, M.(2010) Structure 18: 83-93
- PubMed: 20152155 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.str.2009.10.014
- Primary Citation Related Structures:
3EYZ, 3EZ5 - PubMed Abstract:
High-fidelity DNA polymerases copy DNA rapidly and accurately by adding correct deoxynucleotide triphosphates to a growing primer strand of DNA. Following nucleotide incorporation, a series of conformational changes translocate the DNA substrate by one base pair step, readying the polymerase for the next round of incorporation. Molecular dynamics simulations indicate that the translocation consists globally of a polymerase fingers-opening transition, followed by the DNA displacement and the insertion of the template base into the preinsertion site. They also show that the pyrophosphate release facilitates the opening transition and that the universally conserved Y714 plays a key role in coupling polymerase opening to DNA translocation. The transition involves several metastable intermediates in one of which the O helix is bent in the vicinity of G711. Completion of the translocation appears to require a gating motion of the O1 helix, perhaps facilitated by the presence of G715. These roles are consistent with the high level of conservation of Y714 and the two glycine residues at these positions. It is likely that a corresponding mechanism is applicable to other polymerases.
- Department of Chemistry and Chemical Biology, Harvard University, 12 Oxford Street, Cambridge, MA 02138, USA.
Organizational Affiliation: