The crystal structure of glycosyl hydrolase from Deinococcus radiodurans R1
Chang, C., Hatzos-Skintges, C., Kohler, M., Clancy, S., Joachimiak, A.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Glycosyl hyrolase, family 3 | 351 | Deinococcus radiodurans R1 = ATCC 13939 = DSM 20539 | Mutation(s): 0 Gene Names: DR_1333 | 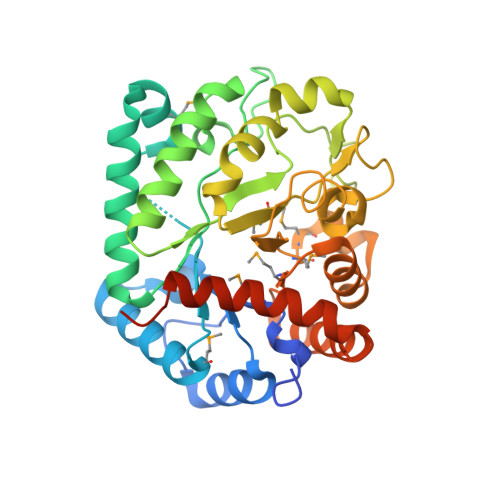 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9RUP9 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| MSE Query on MSE | A, B | L-PEPTIDE LINKING | C5 H11 N O2 Se |  | MET |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 146.841 | α = 90 |
| b = 146.841 | β = 90 |
| c = 108.733 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| PDB_EXTRACT | data extraction |
| SBC-Collect | data collection |
| HKL-3000 | data reduction |
| HKL-3000 | data scaling |
| HKL-3000 | phasing |
| MLPHARE | phasing |
| DM | phasing |
| SHELXDE | phasing |
| RESOLVE | phasing |
| ARP/wARP | model building |
| Coot | model building |