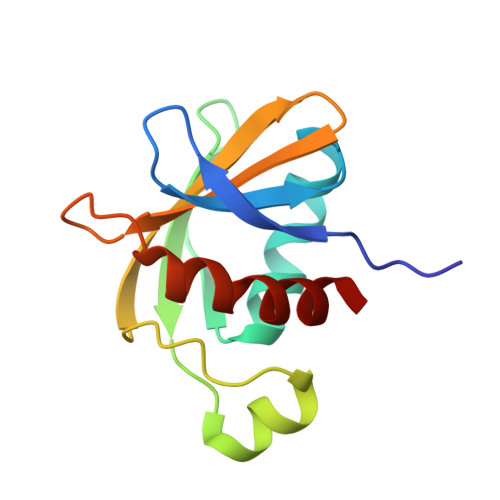
Crystallographic analysis of the C-terminal domain of the Escherichia coli lipoprotein BamC.
Kim, K.H., Aulakh, S., Tan, W., Paetzel, M.(2011) Acta Crystallogr Sect F Struct Biol Cryst Commun 67: 1350-1358
- PubMed: 22102230 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1107/S174430911103363X
- Primary Citation Related Structures:
3SNS - PubMed Abstract:
In Gram-negative bacteria, the BAM complex catalyzes the essential process of assembling outer membrane proteins. The BAM complex in Escherichia coli consists of five proteins: one β-barrel membrane protein, BamA, and four lipoproteins, BamB, BamC, BamD and BamE. Here, the crystal structure of the C-terminal domain of E. coli BamC (BamC(C): Ala224-Ser343) refined to 1.5 Å resolution in space group H3 is reported. BamC(C) consists of a six-stranded antiparallel β-sheet, three α-helices and one 3(10)-helix. Sequence and surface analysis reveals that most of the conserved residues within BamC(C) are localized to form a continuous negatively charged groove that is involved in a major crystalline lattice contact in which a helix from a neighbouring BamC(C) binds against this surface. This interaction is topologically and architecturally similar to those seen in the substrate-binding grooves of other proteins with BamC-like folds. Taken together, these results suggest that an identified surface on the C-terminal domain of BamC may serve as an important protein-binding surface for interaction with other BAM-complex components or substrates.
- Molecular Biology and Biochemistry, Simon Fraser University, 8888 University Drive, Burnaby, BC V5A 1S6, Canada.
Organizational Affiliation: