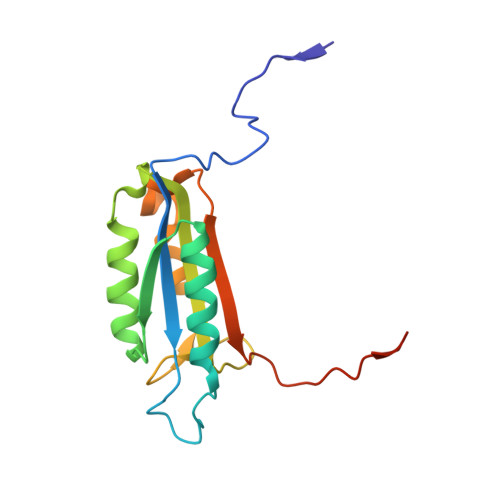
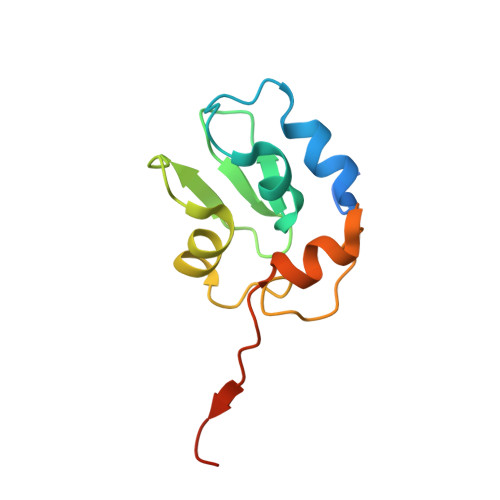
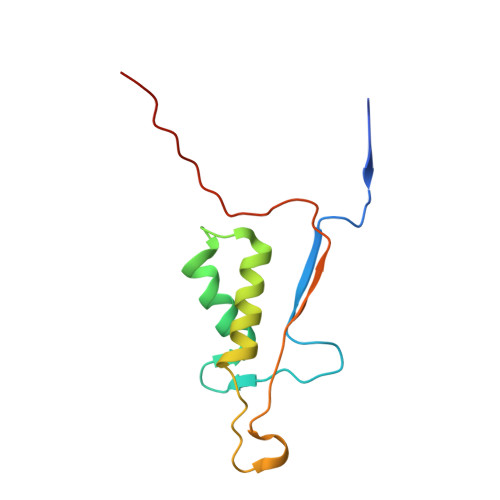
Structural mechanisms of DIAP1 auto-inhibition and DIAP1-mediated inhibition of drICE.
Li, X., Wang, J., Shi, Y.(2011) Nat Commun 2: 408-408
- PubMed: 21811237 Search on PubMed
- DOI: https://doi.org/10.1038/ncomms1418
- Primary Citation Related Structures:
3SIP, 3SIQ, 3SIR - PubMed Abstract:
The Drosophila inhibitor of apoptosis protein DIAP1 exists in an auto-inhibited conformation, unable to suppress the effector caspase drICE. Auto-inhibition is disabled by caspase-mediated cleavage of DIAP1 after Asp20. The cleaved DIAP1 binds to mature drICE, inhibits its protease activity, and, presumably, also targets drICE for ubiquitylation. DIAP1-mediated suppression of drICE is effectively antagonized by the pro-apoptotic proteins Reaper, Hid, and Grim (RHG). Despite rigorous effort, the molecular mechanisms behind these observations are enigmatic. Here we report a 2.4 Å crystal structure of uncleaved DIAP1-BIR1, which reveals how the amino-terminal sequences recognize a conserved surface groove in BIR1 to achieve auto-inhibition, and a 3.5 Å crystal structure of active drICE bound to cleaved DIAP1-BIR1, which provides a structural explanation to DIAP1-mediated inhibition of drICE. These structures and associated biochemical analyses, together with published reports, define the molecular determinants that govern the interplay among DIAP1, drICE and the RHG proteins.
- Ministry of Education Protein Science Laboratory, Center for Structural Biology, School of Life Sciences and School of Medicine, Tsinghua University, Beijing, China.
Organizational Affiliation: