Cellobiose phosphorylase: reconstructing the structural itinerary along the catalytic pathway
Van Hoorebeke, A., Stout, J., Soetaert, W., Van Beeumen, J., Desmet, T., Savvides, S.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Cellobiose phosphorylase | 822 | Cellulomonas uda | Mutation(s): 0 EC: 2.4.1.20 | 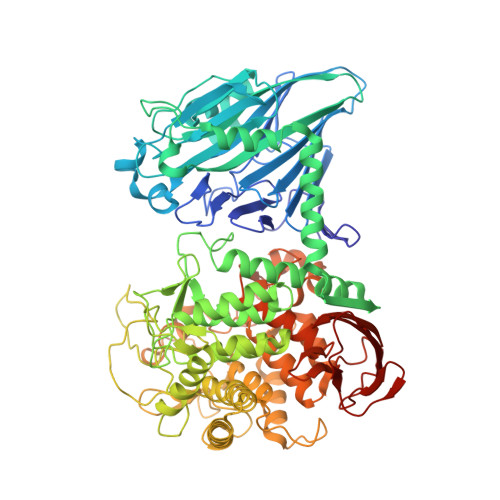 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q7WTR6 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| ID | Chains | Name | Type/Class | 2D Diagram | 3D Interactions |
| PRD_900005 Query on PRD_900005 | C, D | beta-cellobiose | Oligosaccharide / Metabolism |  |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 85.69 | α = 90 |
| b = 195.56 | β = 90 |
| c = 103.64 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| PDB_EXTRACT | data extraction |
| PROTEUM2 | data collection |
| PROTEUM2 | data reduction |
| PROTEUM2 | data scaling |
| PHASER | phasing |