Structural analysis of an archaeal lipoylation system. A bi-partite lipoate protein ligase and its E2 lipoyl domain from Thermoplasma acidophilum
Posner, M.G., Upadhyay, A., Crennell, S., Danson, M.J., Bagby, S.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Lipoate-protein ligase A subunit 1 | 285 | Thermoplasma acidophilum DSM 1728 | Mutation(s): 0 Gene Names: lplA, Ta0514 EC: 2.7.7.63 (PDB Primary Data), 6.3.1.20 (UniProt) | 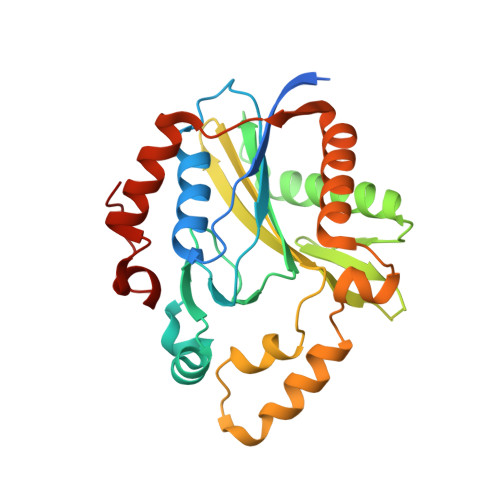 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9HKT1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Putative lipoate-protein ligase A subunit 2 | B [auth C] | 91 | Thermoplasma acidophilum DSM 1728 | Mutation(s): 0 Gene Names: Ta0513, Ta0513m EC: 2.7.7.63 (PDB Primary Data), 6.3.1.20 (UniProt) | 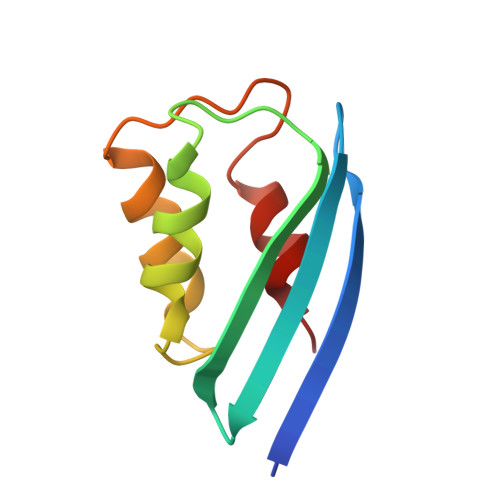 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9HKT2 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| MPD Download:Ideal Coordinates CCD File | C [auth A], D [auth A] | (4S)-2-METHYL-2,4-PENTANEDIOL C6 H14 O2 SVTBMSDMJJWYQN-YFKPBYRVSA-N |  | ||
| ACT Download:Ideal Coordinates CCD File | E [auth A], F [auth C] | ACETATE ION C2 H3 O2 QTBSBXVTEAMEQO-UHFFFAOYSA-M |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 118.57 | α = 90 |
| b = 118.57 | β = 90 |
| c = 72.92 | γ = 120 |
| Software Name | Purpose |
|---|---|
| ADSC | data collection |
| BALBES | phasing |
| REFMAC | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |