Structure and activity of a DUF89 protein from Saccharomyces cerevisiae revealed a novel family of carbohydrate phosphatases
Brown, G., Xu, X., Petit, P., Cui, H., Flick, R., Cuff, M., Savchenko, A., Yakunin, A.F.To be published.
Experimental Data Snapshot
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| UPF0364 protein YMR027W | 471 | Saccharomyces cerevisiae | Mutation(s): 0 Gene Names: YM9711.17, YMR027W EC: 3.1.3 | 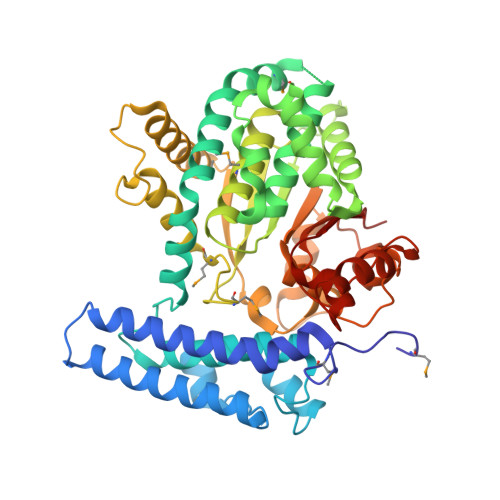 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q04371 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| F6P Download:Ideal Coordinates CCD File | C [auth A] | 6-O-phosphono-beta-D-fructofuranose C6 H13 O9 P BGWGXPAPYGQALX-ARQDHWQXSA-N |  | ||
| GOL Download:Ideal Coordinates CCD File | B [auth A] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| MG Download:Ideal Coordinates CCD File | D [auth A] | MAGNESIUM ION Mg JLVVSXFLKOJNIY-UHFFFAOYSA-N |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| MSE Query on MSE | A | L-PEPTIDE LINKING | C5 H11 N O2 Se |  | MET |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 53.32 | α = 90 |
| b = 108.65 | β = 96.84 |
| c = 88.08 | γ = 90 |
| Software Name | Purpose |
|---|---|
| CrystalClear | data collection |
| MOLREP | phasing |
| PHENIX | refinement |
| MOSFLM | data reduction |
| SCALA | data scaling |