Axial Ligand Swapping In Double Mutant Maintains Intradiol-cleavage Chemistry in Protocatechuate 3,4-Dioxygenase
Purpero, V.M., Lipscomb, J.D.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Protocatechuate 3,4-dioxygenase alpha chain | 200 | Pseudomonas putida | Mutation(s): 0 Gene Names: pcaG, pcaH EC: 1.13.11.3 | 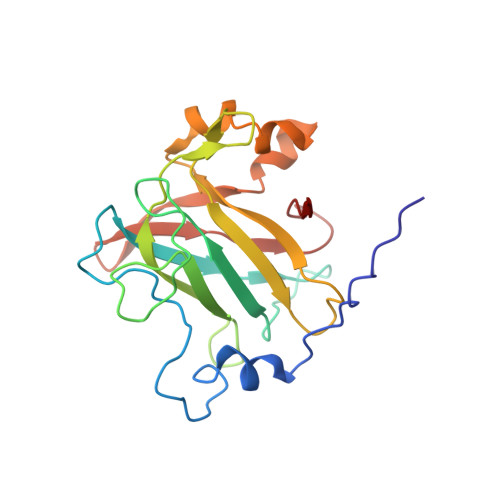 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P00436 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Protocatechuate 3,4-dioxygenase beta chain | D [auth M], E [auth N], F [auth O] | 238 | Pseudomonas putida | Mutation(s): 2 Gene Names: pcaH EC: 1.13.11.3 | 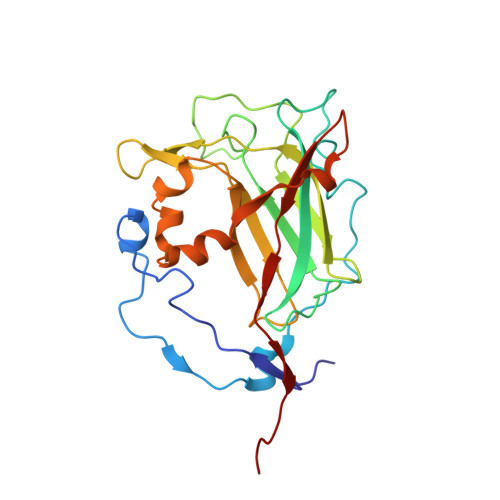 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P00437 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 5 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| SO4 Download:Ideal Coordinates CCD File | G [auth A] GA [auth O] J [auth B] L [auth C] N [auth M] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| GOL Download:Ideal Coordinates CCD File | AA [auth N] H [auth A] K [auth B] M [auth C] O [auth M] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| BME Download:Ideal Coordinates CCD File | EA [auth N] FA [auth N] I [auth A] JA [auth O] KA [auth O] | BETA-MERCAPTOETHANOL C2 H6 O S DGVVWUTYPXICAM-UHFFFAOYSA-N |  | ||
| CO3 Download:Ideal Coordinates CCD File | BA [auth N], CA [auth N], DA [auth N], IA [auth O], V [auth M] | CARBONATE ION C O3 BVKZGUZCCUSVTD-UHFFFAOYSA-L |  | ||
| FE Download:Ideal Coordinates CCD File | HA [auth O], P [auth M], Z [auth N] | FE (III) ION Fe VTLYFUHAOXGGBS-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 128.311 | α = 90 |
| b = 140.487 | β = 90 |
| c = 168.061 | γ = 90 |
| Software Name | Purpose |
|---|---|
| DENZO | data reduction |
| SCALEPACK | data scaling |
| MOLREP | phasing |
| REFMAC | refinement |
| PDB_EXTRACT | data extraction |