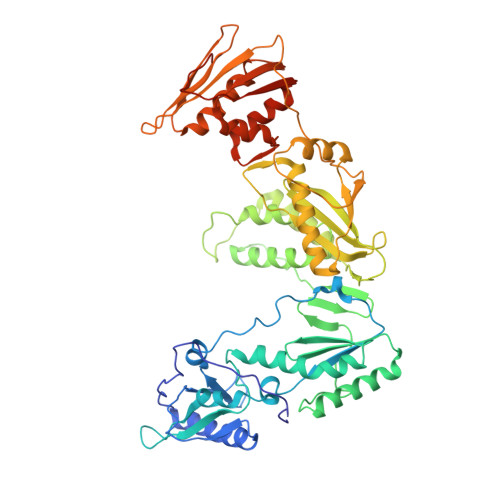
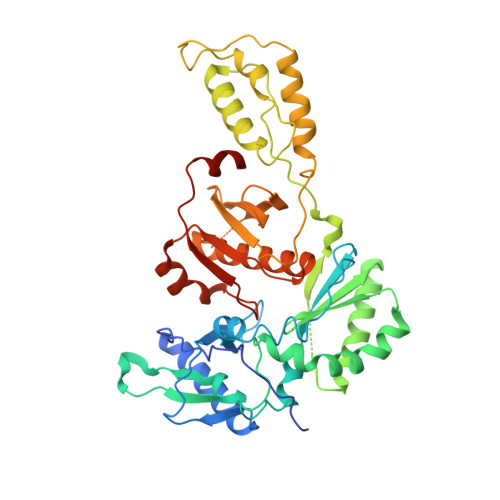
Substituted tetrahydroquinolines as potent allosteric inhibitors of reverse transcriptase and its key mutants.
Su, D.S., Lim, J.J., Tinney, E., Wan, B.L., Young, M.B., Anderson, K.D., Rudd, D., Munshi, V., Bahnck, C., Felock, P.J., Lu, M., Lai, M.T., Touch, S., Moyer, G., Distefano, D.J., Flynn, J.A., Liang, Y., Sanchez, R., Prasad, S., Yan, Y., Perlow-Poehnelt, R., Torrent, M., Miller, M., Vacca, J.P., Williams, T.M., Anthony, N.J.(2009) Bioorg Med Chem Lett 19: 5119-5123
- PubMed: 19631528 Search on PubMed
- DOI: https://doi.org/10.1016/j.bmcl.2009.07.031
- Primary Citation Related Structures:
3I0R, 3I0S - PubMed Abstract:
Non-nucleoside reverse transcriptase inhibitors (NNRTIs) are key elements of multidrug regimens, called HAART (Highly Active Antiretroviral Therapy), that are used to treat HIV-1 infections. Elucidation of the structure-activity relationships of the thiocarbamate moiety of the previous published lead compound 2 provided a series of novel tetrahydroquinoline derivatives as potent inhibitors of HIV-1 RT with nanomolar intrinsic activity on the WT and key mutant enzymes and potent antiviral activity in infected cells. The SAR optimization, mutation profiles, preparation of compounds, and pharmacokinetic profile of compounds are described.
- Department of Medicinal Chemistry and Structural Biology, Merck Research Laboratory, West Point, PA 19486, USA. Dai-Shi.X.Su@gsk.com
Organizational Affiliation: