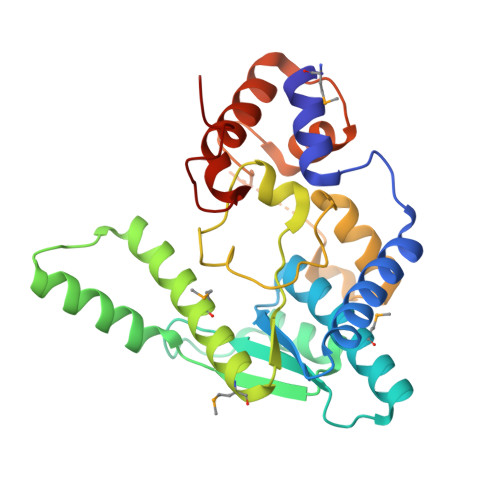
1.9 Angstrom resolution crystal structure of a NAD synthetase (nadE) from Salmonella typhimurium LT2 in complex with NAD(+)
Halavaty, A.S., Wawrzak, Z., Skarina, T., Onopriyenko, O., Peterson, S.N., Savchenko, A., Anderson, W.F., Center for Structural Genomics of Infectious Diseases (CSGID)To be published.