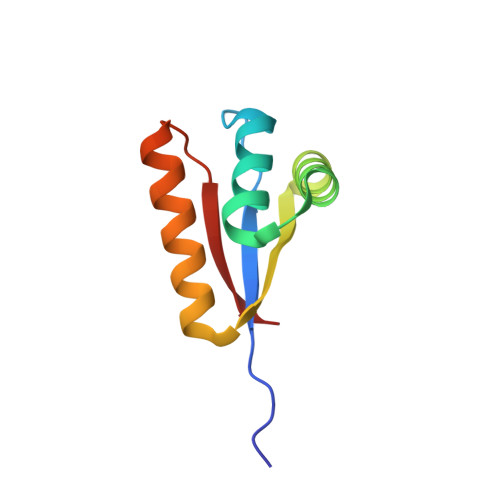
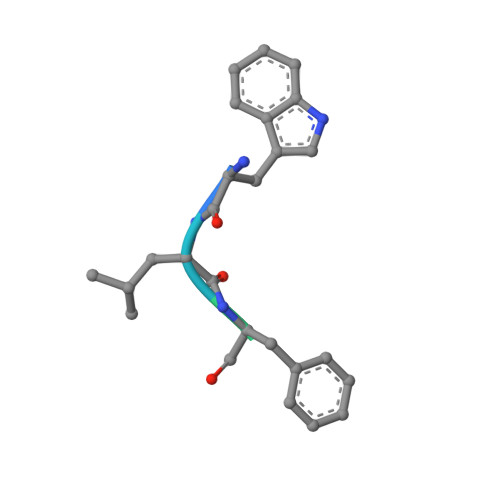
Molecular basis of substrate selection by the N-end rule adaptor protein ClpS.
Roman-Hernandez, G., Grant, R.A., Sauer, R.T., Baker, T.A.(2009) Proc Natl Acad Sci U S A 106: 8888-8893
- PubMed: 19451643 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.0903614106
- Primary Citation Related Structures:
3G19, 3G1B, 3GQ0, 3GQ1, 3GW1 - PubMed Abstract:
The N-end rule is a conserved degradation pathway that relates the stability of a protein to its N-terminal amino acid. Here, we present crystal structures of ClpS, the bacterial N-end rule adaptor, alone and engaged with peptides containing N-terminal phenylalanine, leucine, and tryptophan. These structures, together with a previous structure of ClpS bound to an N-terminal tyrosine, illustrate the molecular basis of recognition of the complete set of primary N-end rule amino acids. In each case, the alpha-amino group and side chain of the N-terminal residue are the major determinants of recognition. The binding pocket for the N-end residue is preformed in the free adaptor, and only small adjustments are needed to accommodate N-end rule residues having substantially different sizes and shapes. M53A ClpS is known to mediate degradation of an expanded repertoire of substrates, including those with N-terminal valine or isoleucine. A structure of Met53A ClpS engaged with an N-end rule tryptophan reveals an essentially wild-type mechanism of recognition, indicating that the Met(53) side chain directly enforces specificity by clashing with and excluding beta-branched side chains. Finally, experimental and structural data suggest mechanisms that make proteins with N-terminal methionine bind very poorly to ClpS, explaining why these high-abundance proteins are not degraded via the N-end rule pathway in the cell.
- Department of Biology, Massachusetts Institute of Technology, Cambridge, MA 02139, USA.
Organizational Affiliation: