Crystal structure of nicotinate-nucleotide pyrophosphorylase from Burkholderi pseudomallei
Seattle Structural Genomics Center for Infectious Disease (SSGCID)To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| nicotinate-nucleotide diphosphorylase (carboxylating) | 298 | Burkholderia pseudomallei 1710b | Mutation(s): 0 Gene Names: nadC, BURPS1710b_1132 EC: 2.4.2.19 | 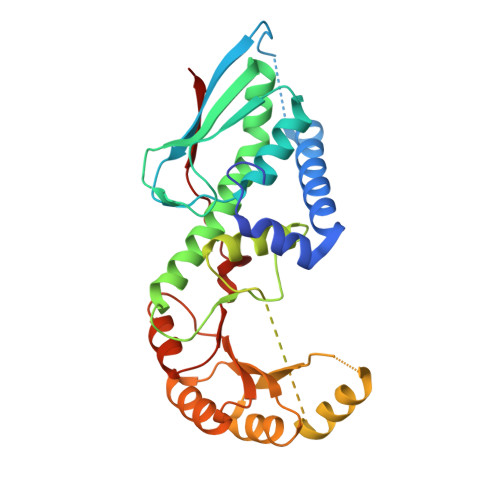 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q3JV59 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Unknown Peptide | C [auth D], D [auth E] | 4 | unidentified | Mutation(s): 0 |  |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 160.978 | α = 90 |
| b = 57.414 | β = 106.74 |
| c = 66.243 | γ = 90 |
| Software Name | Purpose |
|---|---|
| DENZO | data reduction |
| SCALEPACK | data scaling |
| MOLREP | phasing |
| REFMAC | refinement |
| PDB_EXTRACT | data extraction |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |