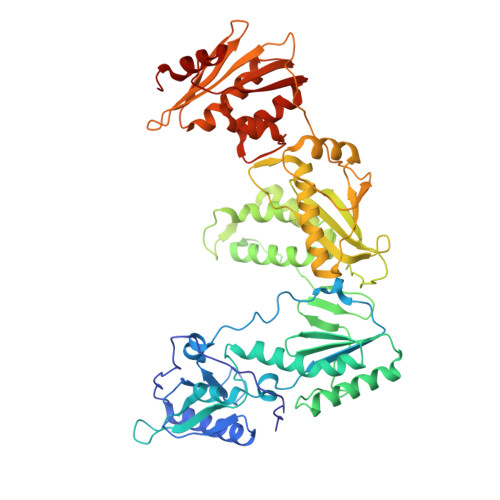
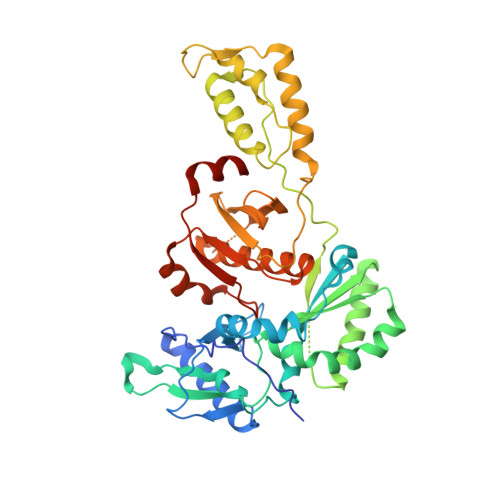
The design and synthesis of diaryl ether second generation HIV-1 non-nucleoside reverse transcriptase inhibitors (NNRTIs) with enhanced potency versus key clinical mutations.
Tucker, T.J., Saggar, S., Sisko, J.T., Tynebor, R.M., Williams, T.M., Felock, P.J., Flynn, J.A., Lai, M.T., Liang, Y., McGaughey, G., Liu, M., Miller, M., Moyer, G., Munshi, V., Perlow-Poehnelt, R., Prasad, S., Sanchez, R., Torrent, M., Vacca, J.P., Wan, B.L., Yan, Y.(2008) Bioorg Med Chem Lett 18: 2959-2966
- PubMed: 18396399 Search on PubMed
- DOI: https://doi.org/10.1016/j.bmcl.2008.03.064
- Primary Citation Related Structures:
3C6T, 3C6U - PubMed Abstract:
Using a combination of traditional Medicinal Chemistry/SAR analysis, crystallography, and molecular modeling, we have designed and synthesized a series of novel, highly potent NNRTIs that possess broad antiviral activity against a number of key clinical mutations.
- Department of Medicinal Chemistry, Merck Research Laboratories, WP14-3, 770 Sumneytown Pike, PO Box 4, West Point, PA 19486-0004, USA. tom_tucker@merck.com
Organizational Affiliation: