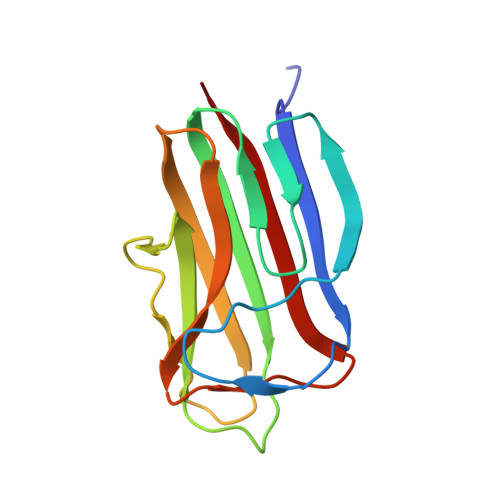
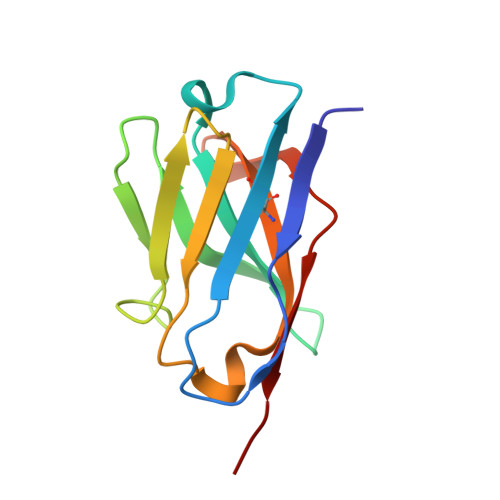
Isolation, kinetic analysis, and structural characterization of an antibody targeting the Bacillus anthracis major spore surface protein BclA.
Nuttall, S.D., Wilkins, M.L., Streltsov, V.A., Pontes-Braz, L., Dolezal, O., Tran, H., Liu, C.Q.(2011) Proteins 79: 1306-1317
- PubMed: 21322055 Search on PubMed
- DOI: https://doi.org/10.1002/prot.22971
- Primary Citation Related Structures:
3AB0 - PubMed Abstract:
One method of laboratory- or field-based testing for anthrax is detection of Bacillus anthracis spores by high-affinity, high specificity binding reagents. From a pool of monoclonal antibodies, we selected one such candidate (A4D11) with high affinity for tBclA, a truncated version of the B. anthracis exosporium protein BclA. Kinetic analysis utilising both standard and kinetic titration on a Biacore biosensor indicated antibody affinities in the 300 pM range for recombinant tBclA, and the A4D11 antibody was also re-formatted into scFv configuration with no loss of affinity. However, assays against B. anthracis and related Bacilli species showed limited binding of intact spores as well as significant cross-reactivity between species. These results were rationalized by determination of the three-dimensional crystallographic structure of the scFv-tBclA complex. A4D11 binds the side of the tBclA trimer, contacting a face of the antigen normally packed against adjacent trimers within the exosporium structure; this inter-spore interface is highly conserved between Bacilli species. Our results indicate the difficulty of generating a high-affinity antibody to differentiate between the highly conserved spore structures of closely related species, but suggest the possibility of future structure-based antibody design for this difficult target.
- CSIRO Division of Materials Science and Engineering, Parkville, Victoria, 3052, Australia. Stewart.Nuttall@csiro.au
Organizational Affiliation: