Structure-reactivity correlations and kinetic isotope effects in aromatic amine dehydrogenase
Hothi, P., Roujeinikova, A., Sutcliffe, M.J., Cullis, P., Leys, D., Scrutton, N.S.To be published.
Experimental Data Snapshot
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Aromatic amine dehydrogenase, small subunit | A [auth D], B [auth H] | 122 | Alcaligenes faecalis | Mutation(s): 1 EC: 1.4.99.4 (PDB Primary Data), 1.4.9.2 (UniProt) | 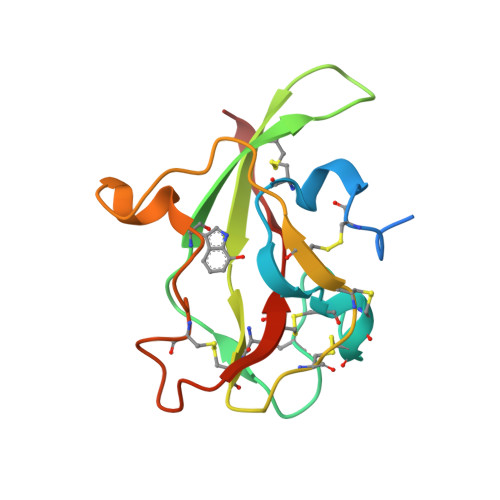 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P84887 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Aromatic amine dehydrogenase, large subunit | C [auth A], D [auth B] | 362 | Alcaligenes faecalis | Mutation(s): 0 Gene Names: aauB EC: 1.4.99.4 (PDB Primary Data), 1.4.9.2 (UniProt) | 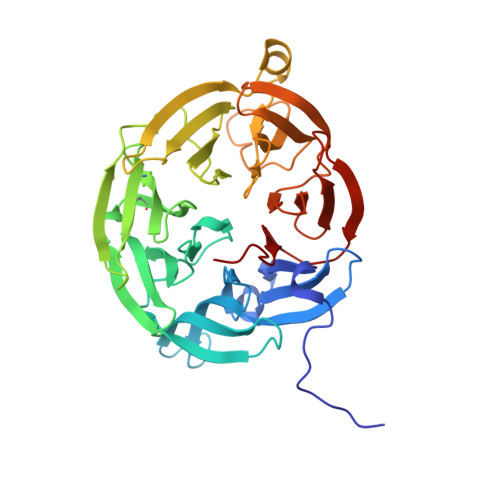 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P84888 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| ZHZ Download:Ideal Coordinates CCD File | F [auth H] | 2-(4-METHOXYPHENYL)ACETAMIDE C9 H11 N O2 OLKQIWCQICCYQS-UHFFFAOYSA-N |  | ||
| ZHH Download:Ideal Coordinates CCD File | E [auth D] | 2-(4-METHOXYPHENYL)ETHANAMINE C9 H13 N O LTPVSOCPYWDIFU-UHFFFAOYSA-N |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| TRQ Query on TRQ | A [auth D], B [auth H] | L-PEPTIDE LINKING | C11 H10 N2 O4 |  | TRP |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 70.848 | α = 90 |
| b = 88.813 | β = 90.39 |
| c = 80.044 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| PDB_EXTRACT | data extraction |
| MOSFLM | data reduction |
| SCALA | data scaling |