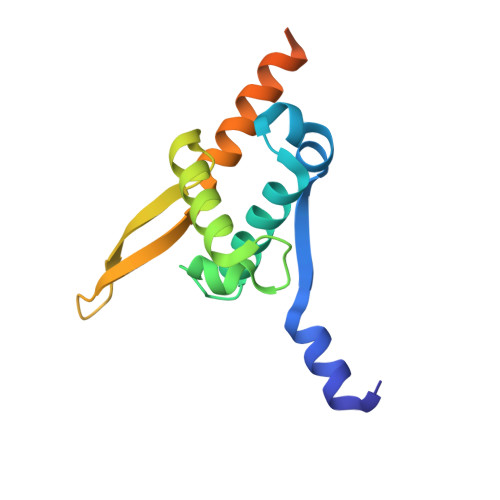
Structure of the N-Terminal Oligomerization Domain of Dnad Reveals a Unique Tetramerization Motif and Provides Insights Into Scaffold Formation.
Schneider, S., Zhang, W., Soultanas, P., Paoli, M.(2008) J Mol Biology 376: 1237
- PubMed: 18206906 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.jmb.2007.12.045
- Primary Citation Related Structures:
2V79 - PubMed Abstract:
DnaD is a primosomal protein that remodels supercoiled plasmids. It binds to supercoiled forms and converts them to open forms without nicking. During this remodeling process, all the writhe is converted to twist and the plasmids are held around the periphery of large scaffolds made up of DnaD molecules. This DNA-remodeling function is the sum of a scaffold-forming activity on the N-terminal domain and a DNA-dependent oligomerization activity on the C-terminal domain. We have determined the crystal structure of the scaffold-forming N-terminal domain, which reveals a winged-helix architecture, with additional structural elements extending from both N- and C-termini. Four monomers form dimers that join into a tetramer. The N-terminal extension mediates dimerization and tetramerization, with extensive interactions and distinct interfaces. The wings and helices of the winged-helix domains remain exposed on the surface of the tetramer. Structure-guided mutagenesis and atomic force microscopy imaging indicate that these elements, together with the C-terminal extension, are involved in scaffold formation. Based upon our data, we propose a model for the DnaD-mediated scaffold formation.
- Centre for Biomolecular Sciences, University of Nottingham, University Park, Nottingham NG7 2RD, UK.
Organizational Affiliation: