Structure of a Novel N-Acyl-Enzyme Intermediate of an N-Terminal Nucleophile (Ntn) Hydrolase, Oat2
Iqbal, A., Schofield, C.J., Clifton, I.J.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| GLUTAMATE N-ACETYLTRANSFERASE 2 ALPHA CHAIN | 173 | Streptomyces clavuligerus | Mutation(s): 0 EC: 2.3.1.35 | 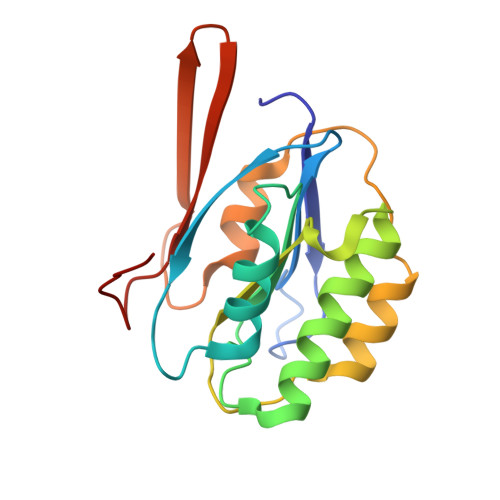 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P0DJQ5 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| GLUTAMATE N-ACETYLTRANSFERASE 2 BETA CHAIN | 213 | Streptomyces clavuligerus | Mutation(s): 1 EC: 2.3.1.35 | 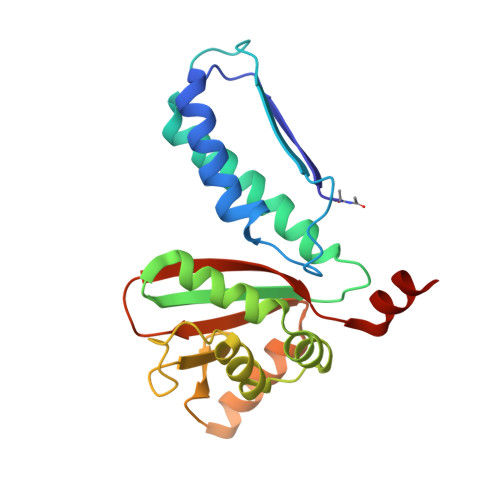 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P0DJQ5 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| AYA Query on AYA | B, D, F, H | L-PEPTIDE LINKING | C5 H9 N O3 |  | ALA |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 73.455 | α = 90 |
| b = 180.163 | β = 90 |
| c = 60.962 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| MOSFLM | data reduction |
| SCALA | data scaling |
| RIGID-BODY | phasing |