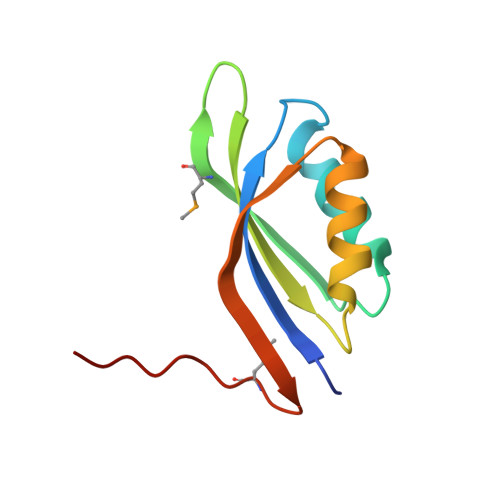
Crystal structure of the MTH889 protein from Methanothermobacter thermautotrophicus.
Forouhar, F., Su, M., Xu, X., Seetharaman, J., Mao, L., Xiao, R., Ma, L.-C., Conover, K., Baran, M.C., Acton, T.B., Montelione, G.T., Arrowsmith, C.H., Hunt, J.F., Tong, L.To be published.