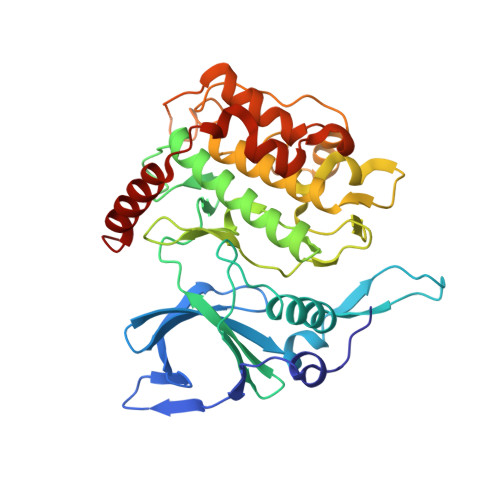
Structures of PfPK7 an atypical protein kinase from P. falciparum identify a novel activation motif and leads for inhibitor design
Merckx, A., Echalier, A., Langford, K., Sicard, A., Langsley, G., Joore, J., Doerig, C., Noble, M., Endicott, J.To be published.