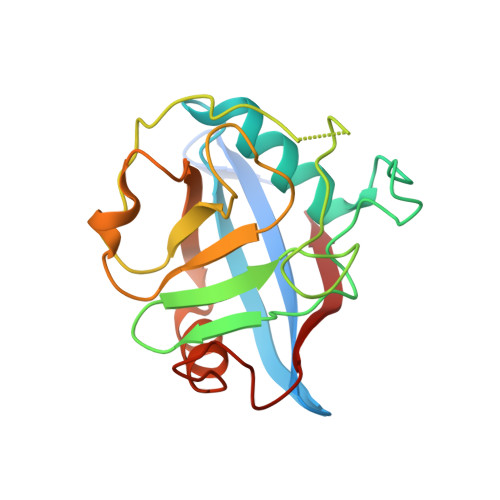
Crystal structure of Cryptosporidium parvum cyclophilin type peptidyl-prolyl cis-trans isomerase cgd2_4120.
Wernimont, A.K., Lew, J., Hills, T., Kozieradzki, I., Lin, Y.H., Hassanali, A., Zhao, Y., Schapira, M., Arrowsmith, C.H., Edwards, A.M., Weigelt, J., Sundstrom, M., Bochkarev, A., Hui, R., Artz, J.D., Xiao, T.To be published.