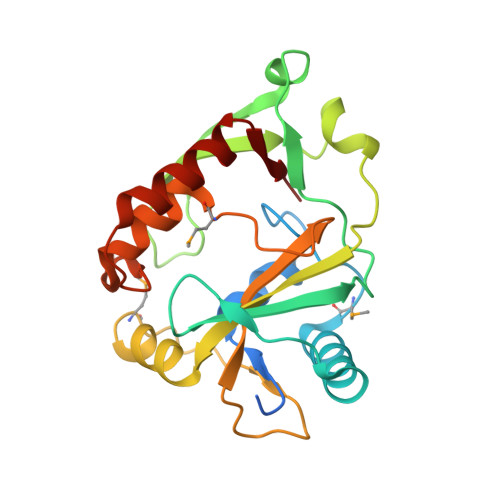
Protein cgd2_2020 from Cryptosporidium parvum
Cymborowski, M., Chruszcz, M., Hills, T., Lew, J., Melone, M., Zhao, Y., Artz, J., Wernimont, A., Edwards, A., Sundstrom, M., Weigelt, J., Bochkarev, A., Arrowsmith, C., Hui, R., Minor, W.To be published.