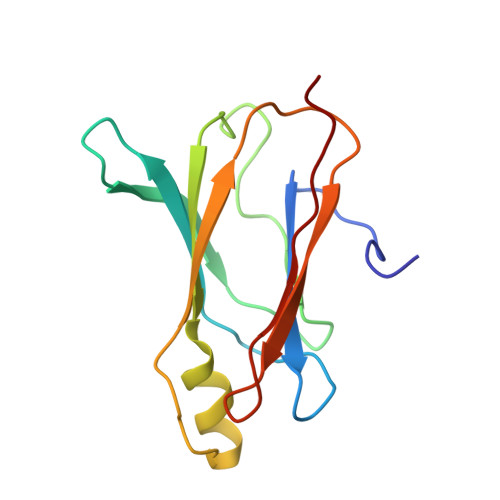
Structure of Monomeric Transthyretin Carrying the Clinically Important T119M Mutation.
Kim, J.H., Oroz, J., Zweckstetter, M.(2016) Angew Chem Int Ed Engl 55: 16168-16171
- PubMed: 27885756 Search on PubMed
- DOI: https://doi.org/10.1002/anie.201608516
- Primary Citation Related Structures:
2NBP - PubMed Abstract:
Mutations in the protein transthyretin can cause as well as protect individuals from transthyretin amyloidosis, an incurable fatal inherited disease. Little is known, however, about the structural basis of pathogenic and clinically protective transthyretin mutants. Here we determined the solution structure of a transthyretin monomer that carries the clinically important T119M mutation. The structure displays a non-native arrangement that is distinct from all known structures of transthyretin and highlights the importance of high-resolution studies in solution for understanding molecular processes that lead to amyloid diseases.
- Deutsches Zentrum für Neurodegenerative Erkrankungen (DZNE), Von-Siebold-Strasse 3a, 37075, Göttingen, Germany.
Organizational Affiliation: