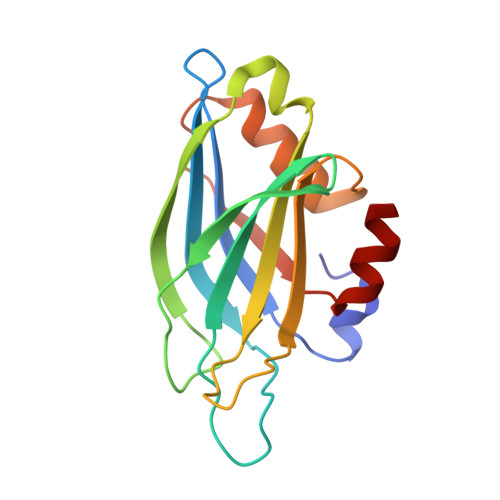
Dynamic binding mode of a Synaptotagmin-1-SNARE complex in solution.
Brewer, K.D., Bacaj, T., Cavalli, A., Camilloni, C., Swarbrick, J.D., Liu, J., Zhou, A., Zhou, P., Barlow, N., Xu, J., Seven, A.B., Prinslow, E.A., Voleti, R., Haussinger, D., Bonvin, A.M., Tomchick, D.R., Vendruscolo, M., Graham, B., Sudhof, T.C., Rizo, J.(2015) Nat Struct Mol Biol 22: 555-564
- PubMed: 26030874 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/nsmb.3035
- Primary Citation Related Structures:
2N1T - PubMed Abstract:
Rapid neurotransmitter release depends on the Ca2+ sensor Synaptotagmin-1 (Syt1) and the SNARE complex formed by synaptobrevin, syntaxin-1 and SNAP-25. How Syt1 triggers release has been unclear, partly because elucidating high-resolution structures of Syt1-SNARE complexes has been challenging. An NMR approach based on lanthanide-induced pseudocontact shifts now reveals a dynamic binding mode in which basic residues in the concave side of the Syt1 C2B-domain β-sandwich interact with a polyacidic region of the SNARE complex formed by syntaxin-1 and SNAP-25. The physiological relevance of this dynamic structural model is supported by mutations in basic residues of Syt1 that markedly impair SNARE-complex binding in vitro and Syt1 function in neurons. Mutations with milder effects on binding have correspondingly milder effects on Syt1 function. Our results support a model whereby dynamic interaction facilitates cooperation between Syt1 and the SNAREs in inducing membrane fusion.
- 1] Department of Biophysics, University of Texas Southwestern Medical Center, Dallas, Texas, USA. [2] Department of Biochemistry, University of Texas Southwestern Medical Center, Dallas, Texas, USA. [3] Department of Pharmacology, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
Organizational Affiliation: