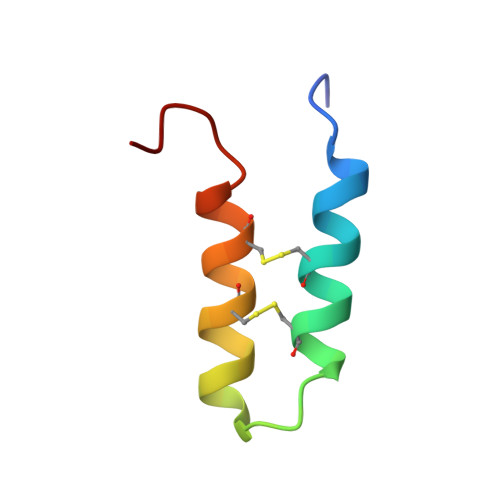
Buckwheat trypsin inhibitor with helical hairpin structure belongs to a new family of plant defence peptides.
Oparin, P.B., Mineev, K.S., Dunaevsky, Y.E., Arseniev, A.S., Belozersky, M.A., Grishin, E.V., Egorov, T.A., Vassilevski, A.A.(2012) Biochem J 446: 69-77
- PubMed: 22612157 Search on PubMed
- DOI: https://doi.org/10.1042/BJ20120548
- Primary Citation Related Structures:
2LQX - PubMed Abstract:
A new peptide trypsin inhibitor named BWI-2c was obtained from buckwheat (Fagopyrum esculentum) seeds by sequential affinity, ion exchange and reversed-phase chromatography. The peptide was sequenced and found to contain 41 amino acid residues, with four cysteine residues involved in two intramolecular disulfide bonds. Recombinant BWI-2c identical to the natural peptide was produced in Escherichia coli in a form of a cleavable fusion with thioredoxin. The 3D (three-dimensional) structure of the peptide in solution was determined by NMR spectroscopy, revealing two antiparallel α-helices stapled by disulfide bonds. Together with VhTI, a trypsin inhibitor from veronica (Veronica hederifolia), BWI-2c represents a new family of protease inhibitors with an unusual α-helical hairpin fold. The linker sequence between the helices represents the so-called trypsin inhibitory loop responsible for direct binding to the active site of the enzyme that cleaves BWI-2c at the functionally important residue Arg(19). The inhibition constant was determined for BWI-2c against trypsin (1.7×10(-1)0 M), and the peptide was tested on other enzymes, including those from various insect digestive systems, revealing high selectivity to trypsin-like proteases. Structural similarity shared by BWI-2c, VhTI and several other plant defence peptides leads to the acknowledgement of a new widespread family of plant peptides termed α-hairpinins.
- M. M. Shemyakin and Yu. A. Ovchinnikov Institute of Bioorganic Chemistry, Russian Academy of Sciences, ul. Miklukho-Maklaya, 16/10, 117997 Moscow, Russia.
Organizational Affiliation: