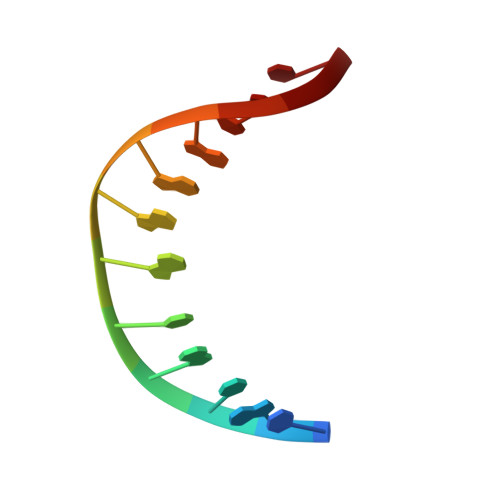
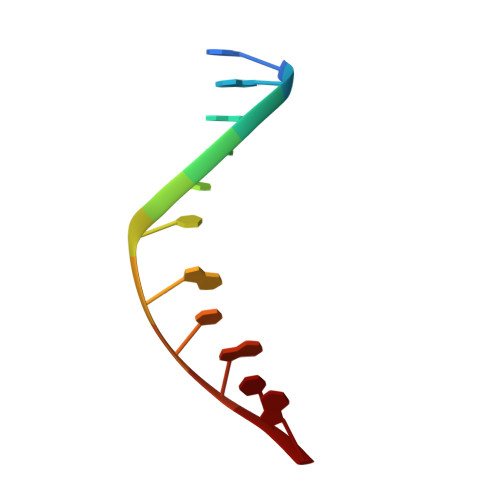
Structure of the 1,N(2)-etheno-2'-deoxyguanosine lesion in the 3'-G(epsilon dG)T-5' sequence opposite a one-base deletion.
Shanmugam, G., Kozekov, I.D., Guengerich, F.P., Rizzo, C.J., Stone, M.P.(2010) Biochemistry 49: 2615-2626
- PubMed: 20201499 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1021/bi901516d
- Primary Citation Related Structures:
2KTP - PubMed Abstract:
The structure of the 1,N(2)-ethenodeoxyguanosine lesion (1,N(2)-epsilondG) has been characterized in 5'-d(CGCATXGAATCC)-3'.5'-d(GGATTCATGCG)-3' (X = 1,N(2)-epsilondG), in which there is no dC opposite the lesion. This duplex (named the 1-BD duplex) models the product of translesion bypass of 1,N(2)-epsilondG by Sulfolobus solfataricus P2 DNA polymerase IV (Dpo4) [Zang, H., Goodenough, A. K., Choi, J. Y., Irimia, A., Loukachevitch, L. V., Kozekov, I. D., Angel, K. C., Rizzo, C. J., Egli, M., and Guengerich, F. P. (2005) J. Biol. Chem. 280, 29750-29764], leading to a one-base deletion. The T(m) of this duplex is 6 degrees C higher than that of the duplex in which dC is present opposite the 1,N(2)-epsilondG lesion and 8 degrees C higher than that of the unmodified 1-BD duplex. Analysis of NOEs between the 1,N(2)-epsilondG imidazole and deoxyribose H1' protons and between the 1,N(2)-epsilondG etheno H6 and H7 protons and DNA protons establishes that 1,N(2)-epsilondG adopts the anti conformation about the glycosyl bond and that the etheno moiety is accommodated within the helix. The resonances of the 1,N(2)-epsilondG H6 and H7 etheno protons shift upfield relative to the monomer 1,N(2)-epsilondG, attributed to ring current shielding, consistent with their intrahelical location. NMR data reveal that Watson-Crick base pairing is maintained at both the 5' and 3' neighbor base pairs. The structure of the 1-BD duplex has been refined using molecular dynamics calculations restrained by NMR-derived distance and dihedral angle restraints. The increased stability of the 1,N(2)-epsilondG lesion in the absence of the complementary dC correlates with the one-base deletion extension product observed during the bypass of the 1,N(2)-epsilondG lesion by the Dpo4 polymerase, suggesting that stabilization of this bulged intermediate may be significant with regard to the biological processing of the lesion.
- Department of Chemistry, Vanderbilt Institute of Chemical Biology, Center in MolecularToxicology, and Vanderbilt-Ingram Cancer Center, Vanderbilt University, Nashville, Tennessee 37235, USA.
Organizational Affiliation: