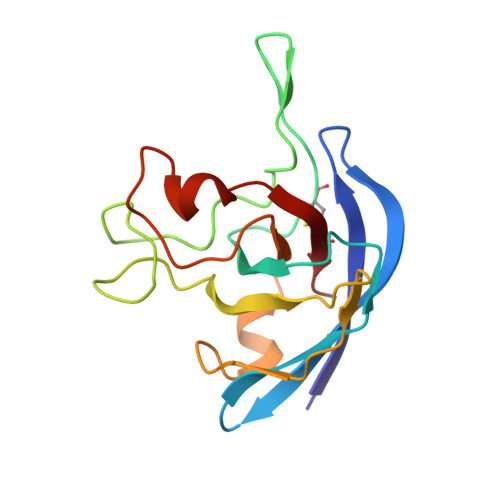
The Solution Structure of the Monomeric Copper, Zinc Superoxide Dismutase from Salmonella enterica: Structural Insights To Understand the Evolution toward the Dimeric Structure.
Mori, M., Jimenez, B., Piccioli, M., Battistoni, A., Sette, M.(2008) Biochemistry 47: 12954-12963
- PubMed: 19006322 Search on PubMed
- DOI: https://doi.org/10.1021/bi801252e
- Primary Citation Related Structures:
2K4W - PubMed Abstract:
The structure of the SodCII-encoded monomeric Cu, Zn superoxide dismutase from Salmonella enterica has been solved by NMR spectroscopy. This represents the first solution structure of a natural and fully active monomeric superoxide dismutase in solution, providing information useful for the interpretation of the evolutional development of these enzymes. The protein scaffold consists of the characteristic beta-barrel common to the whole enzyme family. The general shape of the protein is quite similar to that of Escherichia coli Cu, Zn superoxide dismutase, although some differences are observed mainly in the active site. SodCII presents a more rigid conformation with respect to the engineered monomeric mutants of the human Cu, Zn superoxide dismutase, even though significant disorder is still present in the loops shaping the active site. The analysis of both dynamics and hydration properties of the protein in solution highlights the factors required to maintain the fully active and, at the same time, monomeric protein. This study provides novel insights into the functional differences between monomeric and dimeric bacterial Cu, Zn superoxide dismutases, in turn helping to explain the convergent evolution toward a dimeric structure in prokaryotic and eukaryotic enzymes of this class.
- Magnetic Resonance Center (CERM) and Department of Chemistry, University of Florence, 50019 Sesto Fiorentino (FI), Italy, Department of Biology, University of Rome "Tor Vergata", 00133 Rome, Italy, National Institute of Biostructures and Biosystems (INBB), Viale delle Medaglie d'Oro 305, 00136 Rome, Italy, and Department of Chemical Science and Technology, University of Rome "Tor Vergata", 00133 Rome, Italy e-mail: piccioli@cerm.unifi.it.
Organizational Affiliation: